Chestnut extract induces apoptosis in AGS human gastric cancer cells
- PMID: 21779520
- PMCID: PMC3133749
- DOI: 10.4162/nrp.2011.5.3.185
Chestnut extract induces apoptosis in AGS human gastric cancer cells
Abstract
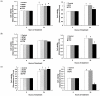
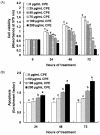
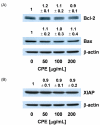
In Korea, chestnut production is increasing each year, but consumption is far below production. We investigated the effect of chestnut extracts on antioxidant activity and anticancer effects. Ethanol extracts of raw chestnut (RCE) or chestnut powder (CPE) had dose-dependent superoxide scavenging activity. Viable numbers of MDA-MD-231 human breast cancer cells, DU145 human prostate cancer cells, and AGS human gastric cancer cells decreased by 18, 31, and 69%, respectively, following treatment with 200 µg/mL CPE for 24 hr. CPE at various concentrations (0-200 µg/mL) markedly decreased AGS cell viability and increased apoptotic cell death dose and time dependently. CPE increased the levels of cleaved caspase-8, -7, -3, and poly (ADP-ribose) polymerase in a dose-dependent manner but not cleaved caspase-9. CPE exerted no effects on Bcl-2 and Bax levels. The level of X-linked inhibitor of apoptosis protein decreased within a narrow range following CPE treatment. The levels of Trail, DR4, and Fas-L increased dose-dependently in CPE-treated AGS cells. These results show that CPE decreases growth and induces apoptosis in AGS gastric cancer cells and that activation of the death receptor pathway contributes to CPE-induced apoptosis in AGS cells. In conclusion, CPE had more of an effect on gastric cancer cells than breast or prostate cancer cells, suggesting that chestnuts would have a positive effect against gastric cancer.
Keywords: Chestnut; anticancer effect; antioxidant activity; ethanol extracts of chestnut; gastric cancer.
Figures


References
-
- Paillet FL. Chestnut: history and ecology of a transformed species. J Biogeogr. 2002;29:1517–1530.
-
- Kim JS, Jung BH, Joo RW, Choi SI. Marketing of chestnut and economic analysis of chestnut cultivation. Korean J For Econ. 2004;12:12–21.
-
- Heo J. Doguibogam. Seoul: Hanmibook press; 2001. p. 1153.
-
- Vekiari SA, Gordon MH, García-Macías P, Labrinea H. Extraction and determination of ellagic acid content in chestnut bark and fruit. Food Chem. 2008;110:1007–1011. - PubMed
-
- Barreira JCM, Ferreira ICFR, Oliveira MBPP, Pereira JA. Antioxidant activities of the extracts from chestnut flower, leaf, skins and fruit. Food Chem. 2008;107:1106–1113.
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

