Enhanced suicidal erythrocyte death in mice carrying a loss-of-function mutation of the adenomatous polyposis coli gene
- PMID: 21781276
- PMCID: PMC4365887
- DOI: 10.1111/j.1582-4934.2011.01387.x
Enhanced suicidal erythrocyte death in mice carrying a loss-of-function mutation of the adenomatous polyposis coli gene
Abstract
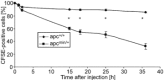
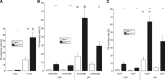
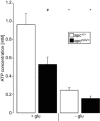
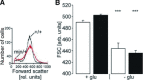
Loss-of-function mutations in human adenomatous polyposis coli (APC) lead to multiple colonic adenomatous polyps eventually resulting in colonic carcinoma. Similarly, heterozygous mice carrying defective APC (apc(Min/+)) suffer from intestinal tumours. The animals further suffer from anaemia, which in theory could result from accelerated eryptosis, a suicidal erythrocyte death triggered by enhanced cytosolic Ca(2+) activity and characterized by cell membrane scrambling and cell shrinkage. To explore, whether APC-deficiency enhances eryptosis, we estimated cell membrane scrambling from annexin V binding, cell size from forward scatter and cytosolic ATP utilizing luciferin-luciferase in isolated erythrocytes from apc(Min/+) mice and wild-type mice (apc(+/+)). Clearance of circulating erythrocytes was estimated by carboxyfluorescein-diacetate-succinimidyl-ester labelling. As a result, apc(Min/+) mice were anaemic despite reticulocytosis. Cytosolic ATP was significantly lower and annexin V binding significantly higher in apc(Min/+) erythrocytes than in apc(+/+) erythrocytes. Glucose depletion enhanced annexin V binding, an effect significantly more pronounced in apc(Min/+) erythrocytes than in apc(+/+) erythrocytes. Extracellular Ca(2+) removal or inhibition of Ca(2+) entry with amiloride (1 mM) blunted the increase but did not abrogate the genotype differences of annexin V binding following glucose depletion. Stimulation of Ca(2+) -entry by treatment with Ca(2+) -ionophore ionomycin (10 μM) increased annexin V binding, an effect again significantly more pronounced in apc(Min/+) erythrocytes than in apc(+/+) erythrocytes. Following retrieval and injection into the circulation of the same mice, apc(Min/+) erythrocytes were more rapidly cleared from circulating blood than apc(+/+) erythrocytes. Most labelled erythrocytes were trapped in the spleen, which was significantly enlarged in apc(Min/+) mice. The observations point to accelerated eryptosis and subsequent clearance of apc(Min/+) erythrocytes, which contributes to or even accounts for the enhanced erythrocyte turnover, anaemia and splenomegaly in those mice.
© 2011 The Authors Journal of Cellular and Molecular Medicine © 2011 Foundation for Cellular and Molecular Medicine/Blackwell Publishing Ltd.
Figures


References
-
- Giles RH, van Es JH, Clevers H. Caught up in a Wnt storm: Wnt signaling in cancer. Biochim Biophys Acta. 2003;1653:1–24. - PubMed
-
- Korinek V, Barker N, Morin PJ, et al. Constitutive transcriptional activation by a beta-catenin-Tcf complex in APC−/− colon carcinoma. Science. 1997;275:1784–7. - PubMed
-
- Morin PJ, Sparks AB, Korinek V, et al. Activation of beta-catenin-Tcf signaling in colon cancer by mutations in beta-catenin or APC. Science. 1997;275:1787–90. - PubMed
-
- Polakis P. Wnt signaling and cancer. Genes Dev. 2000;14:1837–51. - PubMed
-
- He TC, Sparks AB, Rago C, et al. Identification of c-MYC as a target of the APC pathway. Science. 1998;281:1509–12. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

