Signaling by vitamin A and retinol-binding protein in regulation of insulin responses and lipid homeostasis
- PMID: 21782034
- PMCID: PMC3204314
- DOI: 10.1016/j.bbalip.2011.07.002
Signaling by vitamin A and retinol-binding protein in regulation of insulin responses and lipid homeostasis
Abstract
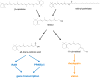
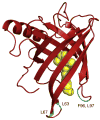
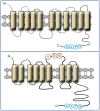
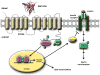
Vitamin A, retinol, circulates in blood bound to serum retinol binding protein (RBP) and is transported into cells by a membrane protein termed stimulated by retinoic acid 6 (STRA6). It was reported that serum levels of RBP are elevated in obese rodents and humans, and that increased level of RBP in blood causes insulin resistance. A molecular mechanism by which RBP can exert such an effect is suggested by the recent discovery that STRA6 is not only a vitamin A transporter but also functions as a surface signaling receptor. Binding of RBP-ROH to STRA6 induces the phosphorylation of a tyrosine residue in the receptor C-terminus, thereby activating a JAK/STAT signaling cascade. Consequently, in STRA6-expressing cells such as adipocytes, RBP-ROH induces the expression of STAT target genes, including SOCS3, which suppresses insulin signaling, and PPARγ, which enhances lipid accumulation. RBP-retinol thus joins the myriad of cytokines, growth factors and hormones which regulate gene transcription by activating cell surface receptors that signal through activation of Janus kinases and their associated transcription factors STATs. This article is part of a Special Issue entitled Retinoid and Lipid Metabolism.
© 2011 Elsevier B.V. All rights reserved.
Figures
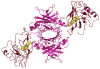
References
-
- McCollum EV, Davis M. The necessity of certain lipins in the diet during growth. J Biol Chem. 1913;15:167–175. - PubMed
-
- Osborne TB, Mendel LB. The vitamins in green foods. J Biol Chem. 1919;37:187–200.
-
- Noy N. Vitamin A. In: Stipanuk MH, editor. Biochemical, physiological, & molecular aspects of human nutrition. 2. Saunders Elsevier; St. Louis: 2006.
-
- Chambon P. A decade of molecular biology of retinoic acid receptors. Faseb J. 1996;10:940–954. - PubMed
-
- Germain P, Chambon P, Eichele G, Evans RM, Lazar MA, Leid M, De Lera AR, Lotan R, Mangelsdorf DJ, Gronemeyer H. International Union of Pharmacology. LX. Retinoic acid receptors. Pharmacol Rev. 2006;58:712–725. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

