Myricetin Ameliorates Defective Post-Receptor Insulin Signaling via β-Endorphin Signaling in the Skeletal Muscles of Fructose-Fed Rats
- PMID: 21785619
- PMCID: PMC3136182
- DOI: 10.1093/ecam/neq017
Myricetin Ameliorates Defective Post-Receptor Insulin Signaling via β-Endorphin Signaling in the Skeletal Muscles of Fructose-Fed Rats
Abstract
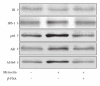
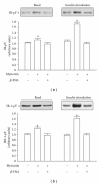
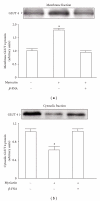
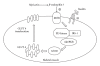
β-Endorphin plays a major role in the amelioration of insulin resistance. The present study documents that myricetin (3,5,7,3', 4', 5'-hexahydroxyflavone) ameliorates insulin resistance by enhancing β-endorphin production in insulin-resistant rats. The rats were induced for insulin resistance by feeding them a diet containing 60% fructose for 6 weeks. The degree of insulin resistance was measured by the homeostasis model assessment of basal insulin resistance (HOMA-IR). The plasma levels of insulin and β-endorphin were measured by an enzyme-linked immunosorbent assay. The insulin receptor-related signaling mediators in the soleus muscles of rats were evaluated by immunoprecipitation or immunoblotting. Myricetin was injected daily (1 mg kg(-1) per injection, thrice daily) for 14 days. Consequently, the high-glucose plasma levels in fructose-fed rats decreased significantly concomitant with an increase in plasma β-endorphin. The reduction of the elevated HOMA-IR index following treatment with myricetin was subsequently inhibited by the administration of β-funaltrexamine hydrochloride (β-FNA) at doses sufficient to block μ-opioid receptors (MOR). The myricetin treatment was also observed to affect the phosphorylation of the insulin receptor, insulin receptor substrate-1, Akt and Akt substrate of 160 kDa, with subsequent effects on glucose-transporter subtype 4 translocation, all of which were blocked by β-FNA pretreatment. These results indicated that enhancement of β-endorphin secretion, which in turn leads to peripheral MOR activation, is involved in the action of myricetin on the amelioration of impaired signaling intermediates downstream of insulin receptors.
Figures


References
-
- Ong KC, Khoo H-E. Biological effects of myricetin. General Pharmacology. 1997;29(2):121–126. - PubMed
-
- Ko C-H, Shen S-C, Lee TJF, Chen Y-C. Myricetin inhibits matrix metalloproteinase 2 protein expression and enzyme activity in colorectal carcinoma cells. Molecular Cancer Therapeutics. 2005;4(2):281–290. - PubMed
-
- Ong KC, Khoo H-E. Insulinomimetic effects of myricetin on lipogenesis and glucose transport in rat adipocytes but not glucose transporter translocation. Biochemical Pharmacology. 1996;51(4):423–429. - PubMed
-
- Ong KC, Khoo H-E. Effects of myricetin on glycemia and glycogen metabolism in diabetic rats. Life Sciences. 2000;67(14):1695–1705. - PubMed
-
- Liu I-M, Liou S-S, Lan T-W, Hsu F-L, Cheng J-T. Myricetin as the active principle of Abelmoschus moschatus to lower plasma glucose in streptozotocin-induced diabetic rats. Planta Medica. 2005;71(7):617–621. - PubMed
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials

