Solid-state NMR spectroscopy provides atomic-level insights into the dehydration of cartilage
- PMID: 21786810
- PMCID: PMC3158280
- DOI: 10.1021/jp205663z
Solid-state NMR spectroscopy provides atomic-level insights into the dehydration of cartilage
Abstract
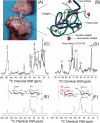
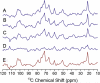
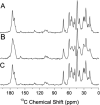
An atomic-level insight into the functioning of articular cartilage would be useful to develop prevention strategies and therapies for joint diseases such as osteoarthritis. However, the composition and structure of cartilage and their relationship to its unique mechanical properties are quite complex and pose tremendous challenges to most biophysical techniques. In this study, we present an investigation of the structure and dynamics of polymeric molecules of articular cartilage using time-resolved solid-state NMR spectroscopy during dehydration. Full-thickness cartilage explants were used in magic-angle spinning experiments to monitor the structural changes of rigid and mobile carbons. Our results reveal that the dehydration reduced the mobility of collagen amino acid residues and carbon sugar ring structures in glycosaminoglycans but had no effect on the trans-Xaa-Pro conformation. Equally interestingly, our results demonstrate that the dehydration effects are reversible, and the molecular structure and mobility are restored upon rehydration.
© 2011 American Chemical Society
Figures



References
-
- Cohen NP, Foster RJ, Mow VC. J. Orthop. Sports Phys. Ther. 1998;28:203–15. - PubMed
-
- Kirk TB, Wilson AS, Stachowiak GW. J. Orthop. Rheumatol. 1993;6:21–28.
-
- Naji L, Kaufmann J, Huster D, Schiller J, Arnold K. Carbohydr. Res. 2000;327:439–46. - PubMed
-
- Huster D, Schiller J, Arnold K. Magn. Reson. Med. 2002;48:624–32. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

