CYP2D plays a major role in berberine metabolism in liver of mice and humans
- PMID: 21787170
- PMCID: PMC5507062
- DOI: 10.3109/00498254.2011.597456
CYP2D plays a major role in berberine metabolism in liver of mice and humans
Abstract
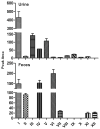
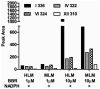
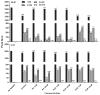
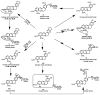
Berberine is a widely used plant extract for gastrointestinal infections, and is reported to have potential benefits in treatment for diabetes and hypercholesterolemia. It has been suggested that interactions between berberine-containing products and cytochromes P450 (CYPs) exist, but little is known about which CYPs mediate the metabolism of berberine in vivo. In this study, berberine metabolites in urine and feces of mice were analyzed, and the role that CYPs play in producing these metabolites were characterized in liver microsomes from mice (MLM) and humans (HLM), as well as recombinant human CYPs. Eleven berberine metabolites were identified in mice, including 5 unconjugated metabolites, mainly in feces, and 6 glucuronide and sulfate conjugates, predominantly in urine. Three novel berberine metabolites were observed. Three unconjugated metabolites of berberine were produced by MLM, HLM, and recombinant human CYPs. CYP2D6 was the primary recombinant human CYP producing these metabolites, followed by CYP1A2, 3A4, 2E1 and CYP2C19. The metabolism of berberine in MLM and HLM was decreased the most by a CYP2D inhibitor, and moderately by inhibitors of CYP1A and 3A. CYP2D plays a major role in berberine biotransformation, therefore, CYP2D6 pharmacogenetics and potential drug-drug interactions should be considered when berberine is used.
Conflict of interest statement
Figures


Similar articles
-
Involvement of CYP3A4/5 and CYP2D6 in the metabolism of aconitine using human liver microsomes and recombinant CYP450 enzymes.Toxicol Lett. 2011 Apr 10;202(1):47-54. doi: 10.1016/j.toxlet.2011.01.019. Epub 2011 Jan 28. Toxicol Lett. 2011. PMID: 21277363
-
Prediction of differences in in vivo oral clearance of N,N-dipropyl-2-[4-methoxy-3-(2-phenylethoxy)phenyl] ethylamine monohydrochloride (NE-100) between extensive and poor metabolizers from in vitro metabolic data in human liver microsomes lacking CYP2D6 activity and recombinant CYPs.Xenobiotica. 2004 Jul;34(7):687-703. doi: 10.1080/00498250412331281070. Xenobiotica. 2004. PMID: 15672756
-
In vitro metabolism of the opioid tilidine and interaction of tilidine and nortilidine with CYP3A4, CYP2C19, and CYP2D6.Naunyn Schmiedebergs Arch Pharmacol. 2008 Sep;378(3):275-82. doi: 10.1007/s00210-008-0294-7. Epub 2008 May 31. Naunyn Schmiedebergs Arch Pharmacol. 2008. PMID: 18516595
-
Identification of human cytochrome P450 isoforms involved in the 7-hydroxylation of chlorpromazine by human liver microsomes.Life Sci. 2000;67(2):175-84. doi: 10.1016/s0024-3205(00)00613-5. Life Sci. 2000. PMID: 10901285
-
In vitro metabolism of l-corydalmine, a potent analgesic drug, in human, cynomolgus monkey, beagle dog, rat and mouse liver microsomes.J Pharm Biomed Anal. 2016 Sep 5;128:98-105. doi: 10.1016/j.jpba.2016.05.024. Epub 2016 May 17. J Pharm Biomed Anal. 2016. PMID: 27239758
Cited by
-
Anti-Inflammatory and Antioxidant Chinese Herbal Medicines: Links between Traditional Characters and the Skin Lipoperoxidation "Western" Model.Antioxidants (Basel). 2022 Mar 23;11(4):611. doi: 10.3390/antiox11040611. Antioxidants (Basel). 2022. PMID: 35453296 Free PMC article. Review.
-
The Quest to Enhance the Efficacy of Berberine for Type-2 Diabetes and Associated Diseases: Physicochemical Modification Approaches.Biomedicines. 2020 Apr 18;8(4):90. doi: 10.3390/biomedicines8040090. Biomedicines. 2020. PMID: 32325761 Free PMC article. Review.
-
Dose-response of berberine on hepatic cytochromes P450 mRNA expression and activities in mice.J Ethnopharmacol. 2011 Oct 31;138(1):111-8. doi: 10.1016/j.jep.2011.08.058. Epub 2011 Sep 6. J Ethnopharmacol. 2011. PMID: 21920422 Free PMC article.
-
Effects of Berberine against Pancreatitis and Pancreatic Cancer.Molecules. 2022 Dec 6;27(23):8630. doi: 10.3390/molecules27238630. Molecules. 2022. PMID: 36500723 Free PMC article. Review.
-
Berberine in Cardiovascular and Metabolic Diseases: From Mechanisms to Therapeutics.Theranostics. 2019 Mar 16;9(7):1923-1951. doi: 10.7150/thno.30787. eCollection 2019. Theranostics. 2019. PMID: 31037148 Free PMC article. Review.
References
-
- Abourashed EA, Khan IA. High-performance liquid chromatography determination of hydrastine and berberine in dietary supplements containing goldenseal. J Pharm Sci. 2001;90:817–822. - PubMed
-
- Amin AH, Subbaiah TV, Abbasi KM. Berberine sulfate: antimicrobial activity, bioassay, and mode of action. Can J Microbiol. 1969;15:1067–1076. - PubMed
-
- Barnes PM, Bloom B, Nahin RL. Complementary and alternative medicine use among adults and children: United States, 2007. Natl Health Stat Report. 2008:1–23. - PubMed
-
- Bogaards JJ, Bertrand M, Jackson P, Oudshoorn MJ, Weaver RJ, van Bladeren PJ, Walther B. Determining the best animal model for human cytochrome P450 activities: a comparison of mouse, rat, rabbit, dog, micropig, monkey and man. Xenobiotica. 2000;30:1131–1152. - PubMed
-
- Deroussent A, Re M, Hoellinger H, Cresteil T. Metabolism of sanguinarine in human and in rat: characterization of oxidative metabolites produced by human CYP1A1 and CYP1A2 and rat liver microsomes using liquid chromatography-tandem mass spectrometry. J Pharm Biomed Anal. 2010;52:391–397. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
