ZEB1 limits adenoviral infectability by transcriptionally repressing the coxsackie virus and adenovirus receptor
- PMID: 21791114
- PMCID: PMC3164624
- DOI: 10.1186/1476-4598-10-91
ZEB1 limits adenoviral infectability by transcriptionally repressing the coxsackie virus and adenovirus receptor
Abstract
Background: We have previously reported that RAS-MEK (Cancer Res. 2003 May 1;63(9):2088-95) and TGF-β (Cancer Res. 2006 Feb 1;66(3):1648-57) signaling negatively regulate coxsackie virus and adenovirus receptor (CAR) cell-surface expression and adenovirus uptake. In the case of TGF-β, down-regulation of CAR occurred in context of epithelial-to-mesenchymal transition (EMT), a process associated with transcriptional repression of E-cadherin by, for instance, the E2 box-binding factors Snail, Slug, SIP1 or ZEB1. While EMT is crucial in embryonic development, it has been proposed to contribute to the formation of invasive and metastatic carcinomas by reducing cell-cell contacts and increasing cell migration.
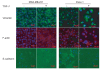
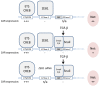
Results: Here, we show that ZEB1 represses CAR expression in both PANC-1 (pancreatic) and MDA-MB-231 (breast) human cancer cells. We demonstrate that ZEB1 physically associates with at least one of two closely spaced and conserved E2 boxes within the minimal CAR promoter here defined as genomic region -291 to -1 relative to the translational start ATG. In agreement with ZEB1's established role as a negative regulator of the epithelial phenotype, silencing its expression in MDA-MB-231 cells induced a partial Mesenchymal-to-Epithelial Transition (MET) characterized by increased levels of E-cadherin and CAR, and decreased expression of fibronectin. Conversely, knockdown of ZEB1 in PANC-1 cells antagonized both the TGF-β-induced down-regulation of E-cadherin and CAR and the reduction of adenovirus uptake. Interestingly, even though ZEB1 clearly contributes to the TGF-β-induced mesenchymal phenotype of PANC-1 cells, TGF-β did not seem to affect ZEB1's protein levels or subcellular localization. These findings suggest that TGF-β may inhibit CAR expression by regulating factor(s) that cooperate with ZEB1 to repress the CAR promoter, rather than by regulating ZEB1 expression levels. In addition to the negative E2 box-mediated regulation the minimal CAR promoter is positively regulated through conserved ETS and CRE elements.
Conclusions: This report provides evidence that inhibition of ZEB1 may improve adenovirus uptake of cancer cells that have undergone EMT and for which ZEB1 is necessary to maintain the mesenchymal phenotype. Targeting of ZEB1 may reverse some aspects of EMT including the down-regulation of CAR.
Figures





References
-
- Lisewski U, Shi Y, Wrackmeyer U, Fischer R, Chen C, Schirdewan A, Juttner R, Rathjen F, Poller W, Radke MH, Gotthardt M. The tight junction protein CAR regulates cardiac conduction and cell-cell communication. The Journal of experimental medicine. 2008;205:2369–2379. doi: 10.1084/jem.20080897. - DOI - PMC - PubMed
-
- Anders M, Christian C, McMahon M, McCormick F, Korn WM. Inhibition of the Raf/MEK/ERK pathway up-regulates expression of the coxsackievirus and adenovirus receptor in cancer cells. Cancer Res. 2003;63:2088–2095. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

