Streptozotocin-induced early thermal hyperalgesia is independent of glycemic state of rats: role of transient receptor potential vanilloid 1(TRPV1) and inflammatory mediators
- PMID: 21794120
- PMCID: PMC3157448
- DOI: 10.1186/1744-8069-7-52
Streptozotocin-induced early thermal hyperalgesia is independent of glycemic state of rats: role of transient receptor potential vanilloid 1(TRPV1) and inflammatory mediators
Abstract
Background: Streptozotocin (STZ) is used as a common tool to induce diabetes and to study diabetes-induced complications including diabetic peripheral neuropathy (DPN). Previously, we have reported that STZ induces a direct effect on neurons through expression and function of the Transient receptor potential vanilloid 1 (TRPV1) channel in sensory neurons resulting in thermal hyperalgesia, even in non-diabetic STZ-treated mice. In the present study, we investigated the role of expression and function of TRPV1 in the central sensory nerve terminals in the spinal cord in STZ-induced hyperalgesia in rats.
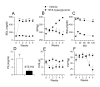
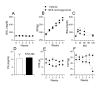
Results: We found that a proportion of STZ-treated rats were normoglycemic but still exhibited thermal hyperalgesia and mechanical allodynia. Immunohistochemical data show that STZ treatment, irrespective of glycemic state of the animal, caused microglial activation and increased expression of TRPV1 in spinal dorsal horn. Further, there was a significant increase in the levels of pro-inflammatory mediators (IL-1β, IL-6 and TNF-α) in spinal cord tissue, irrespective of the glycemic state. Capsaicin-stimulated release of calcitonin gene related peptide (CGRP) was significantly higher in the spinal cord of STZ-treated animals. Intrathecal administration of resiniferatoxin (RTX), a potent TRPV1 agonist, significantly attenuated STZ-induced thermal hyperalgesia, but not mechanical allodynia. RTX treatment also prevented the increase in TRPV1-mediated neuropeptide release in the spinal cord tissue.
Conclusions: From these results, it is concluded that TRPV1 is an integral component of initiating and maintaining inflammatory thermal hyperalgesia, which can be alleviated by intrathecal administration of RTX. Further, the results suggest that enhanced expression and inflammation-induced sensitization of TRPV1 at the spinal cord may play a role in central sensitization in STZ-induced neuropathy.
Figures


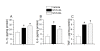


References
-
- Kroncke KD, Fehsel K, Sommer A, Rodriguez ML, Kolb-Bachofen V. Nitric oxide generation during cellular metabolization of the diabetogenic N-methyl-N-nitroso-urea streptozotozin contributes to islet cell DNA damage. Biol Chem. 1995;376:179–185. - PubMed
-
- Roos MD, Wen X, Kaihong S, Clark JA, Xiaoyong Y, Chin E, Paterson AJ, Kudlow JE. Streptozotocin, an analog of N-acetylglucosamine, blocksthe removal of O-GlcNAc from intracellular proteins. Proc Assoc Am Physicians. 1998;110:1–11. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

