Vaccination against heterologous R5 clade C SHIV: prevention of infection and correlates of protection
- PMID: 21799765
- PMCID: PMC3140488
- DOI: 10.1371/journal.pone.0022010
Vaccination against heterologous R5 clade C SHIV: prevention of infection and correlates of protection
Abstract
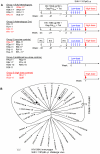
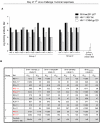
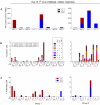
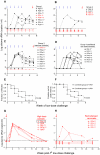
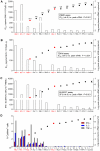
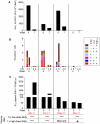
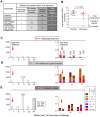
A safe, efficacious vaccine is required to stop the AIDS pandemic. Disappointing results from the STEP trial implied a need to include humoral anti-HIV-1 responses, a notion supported by RV144 trial data even though correlates of protection are unknown. We vaccinated rhesus macaques with recombinant simian immunodeficiency virus (SIV) Gag-Pol particles, HIV-1 Tat and trimeric clade C (HIV-C) gp160, which induced cross-neutralizing antibodies (nAbs) and robust cellular immune responses. After five low-dose mucosal challenges with a simian-human immunodeficiency virus (SHIV) that encoded a heterologous R5 HIV-C envelope (22.1% divergence from the gp160 immunogen), 94% of controls became viremic, whereas one third of vaccinees remained virus-free. Upon high-dose SHIV rechallenge, all controls became infected, whereas some vaccinees remained aviremic. Peak viremia was inversely correlated with both cellular immunity (p<0.001) and cross-nAb titers (p<0.001). These data simultaneously linked cellular as well as humoral immune responses with the degree of protection for the first time.
Conflict of interest statement
Figures
References
-
- Rerks-Ngarm S, Pitisuttithum P, Nitayaphan S, Kaewkungwal J, Chiu J, et al. Vaccination with ALVAC and AIDSVAX to prevent HIV-1 infection in Thailand. N Engl J Med. 2009;361:2209–2220. - PubMed
-
- Koup RA, Graham BS, Douek DC. The quest for a T cell-based immune correlate of protection against HIV: a story of trials and errors. Nat Rev Immunol. 2010;11:65–70. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

