Differential micro-RNA expression in primary CNS and nodal diffuse large B-cell lymphomas
- PMID: 21803762
- PMCID: PMC3177663
- DOI: 10.1093/neuonc/nor107
Differential micro-RNA expression in primary CNS and nodal diffuse large B-cell lymphomas
Abstract
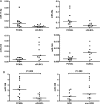
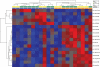
Most primary CNS lymphomas (PCNSL) are diffuse large B-cell lymphomas (DLBCL). However, clinical behavior and prognosis differ considerably from those for nodal DLBCL (nDLBCL), and their pathogenesis is still not fully understood. Micro-RNAs (miRNAs) have been associated with cancer development and progression. We investigated a large miRNA panel for differential expression in PCNSL and nDLBCL, to determine new mechanisms potentially involved in PCNSL pathogenesis. Using paraffin-embedded biopsy specimens from 21 HIV-negative patients with newly diagnosed PCNSL (n = 11) and nDLBCL (n= 10), we measured the expression of 365 miRNA species by quantitative real-time PCR using low-density PCR arrays. We found that 18 miRNAs were differentially expressed: median expression levels of 13 miRNAs were 2.1-13.1 times higher in PCNSL, and median expression levels of 5 miRNAs were 2.6-3.3 times higher in nDLBCL. MiRNAs upregulated in PCNSL were associated with the Myc pathway (miR-17-5p, miR-20a, miR-9), with blocking of terminal B-cell differentiation (miR-9, miR-30b/c), or with upregulation by inflammatory cytokines (miR-155). Putative tumor-suppressor miRNAs (miR-199a, miR-214, miR-193b, miR-145) were downregulated in PCNSL. There was no overlap of miRNAs dysregulated in PCNSL with those differentially expressed between immunohistologically defined germinal center B cell-like (GCB) and non-GCB types or, apart from miR-9, with miRNAs known to be overexpressed in human brain. We conclude that PCNSL exhibits a distinct pattern of miRNA expression compared with nDLBCL. This argues for the involvement of different molecular mechanisms in the pathogenesis of these two lymphoma types.
Figures



Similar articles
-
Specific expression of miR-17-5p and miR-127 in testicular and central nervous system diffuse large B-cell lymphoma.Mod Pathol. 2009 Apr;22(4):547-55. doi: 10.1038/modpathol.2009.10. Epub 2009 Mar 13. Mod Pathol. 2009. PMID: 19287466
-
Identification of microRNAs in the cerebrospinal fluid as marker for primary diffuse large B-cell lymphoma of the central nervous system.Blood. 2011 Mar 17;117(11):3140-6. doi: 10.1182/blood-2010-09-308684. Epub 2011 Jan 3. Blood. 2011. PMID: 21200023
-
[Significance of microRNA-146b-5p in diffuse large B-cell lymphoma and its relationship with risk assessment].Zhonghua Xue Ye Xue Za Zhi. 2012 Dec;33(12):1010-4. Zhonghua Xue Ye Xue Za Zhi. 2012. PMID: 23363792 Chinese.
-
Systems biology of primary CNS lymphoma: from genetic aberrations to modeling in mice.Acta Neuropathol. 2014 Feb;127(2):175-88. doi: 10.1007/s00401-013-1202-x. Epub 2013 Nov 16. Acta Neuropathol. 2014. PMID: 24240734 Review.
-
Pathology and new insights in central nervous system lymphomas.Curr Opin Oncol. 2023 Sep 1;35(5):347-356. doi: 10.1097/CCO.0000000000000978. Epub 2023 Jul 13. Curr Opin Oncol. 2023. PMID: 37439536 Free PMC article. Review.
Cited by
-
Restoration of microRNA-214 expression reduces growth of myeloma cells through positive regulation of P53 and inhibition of DNA replication.Haematologica. 2013 Apr;98(4):640-8. doi: 10.3324/haematol.2012.070011. Epub 2012 Oct 25. Haematologica. 2013. PMID: 23100276 Free PMC article.
-
Beyond high-dose methotrexate and brain radiotherapy: novel targets and agents for primary CNS lymphoma.Ann Oncol. 2014 Feb;25(2):316-22. doi: 10.1093/annonc/mdt385. Epub 2013 Nov 20. Ann Oncol. 2014. PMID: 24265352 Free PMC article. Review.
-
Role of microRNAs in primary central nervous system lymphomas.Cell Prolif. 2016 Apr;49(2):147-53. doi: 10.1111/cpr.12243. Epub 2016 Mar 16. Cell Prolif. 2016. PMID: 26990358 Free PMC article. Review.
-
The Challenge of Primary Central Nervous System Lymphoma.Hematol Oncol Clin North Am. 2016 Dec;30(6):1293-1316. doi: 10.1016/j.hoc.2016.07.013. Hematol Oncol Clin North Am. 2016. PMID: 27888882 Free PMC article. Review.
-
Deregulation mechanisms and therapeutic opportunities of p53-responsive microRNAs in diffuse large B-cell lymphoma.PeerJ. 2025 Jan 7;13:e18661. doi: 10.7717/peerj.18661. eCollection 2025. PeerJ. 2025. PMID: 39802185 Free PMC article. Review.
References
-
- Miller DC, Hochberg FH, Harris NL, et al. Pathology with clinical correlations of primary central nervous system non-Hodgkin's lymphoma. The Massachusetts General Hospital experience 1958–1989. Cancer. 1994;74:1383–1397. doi:10.1002/1097-0142(19940815)74:4<1383::AID-CNCR2820740432>3.0.C.... - DOI - PubMed
-
- DeAngelis LM, Seiferheld W, Schold SC, Fisher B, Schultz CJ. Combination chemotherapy and radiotherapy for primary central nervous system lymphoma: Radiation Therapy Oncology Group Study 93-10. J Clin Oncol. 2002;20:4643–4648. doi:10.1200/JCO.2002.11.013. - DOI - PubMed
-
- Calin GA, Croce CM. MicroRNA signatures in human cancers. Nat Rev Cancer. 2006;6:857–866. doi:10.1038/nrc1997. - DOI - PubMed
-
- Bartel DP. MicroRNAs: genomics, biogenesis, mechanism, and function. Cell. 2004;116:281–297. doi:10.1016/S0092-8674(04)00045-5. - DOI - PubMed
-
- Iorio MV, Croce CM. MicroRNAs in cancer: small molecules with a huge impact. J Clin Oncol. 2009;27:5848–5856. doi:10.1200/JCO.2009.24.0317. - DOI - PMC - PubMed

