Role of glucosyltransferase B in interactions of Candida albicans with Streptococcus mutans and with an experimental pellicle on hydroxyapatite surfaces
- PMID: 21803906
- PMCID: PMC3187131
- DOI: 10.1128/AEM.05203-11
Role of glucosyltransferase B in interactions of Candida albicans with Streptococcus mutans and with an experimental pellicle on hydroxyapatite surfaces
Abstract
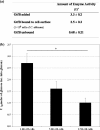
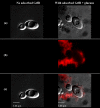
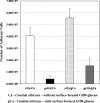
Candida albicans and mutans streptococci are frequently detected in dental plaque biofilms from toddlers afflicted with early childhood caries. Glucosyltransferases (Gtfs) secreted by Streptococcus mutans bind to saliva-coated apatite (sHA) and to bacterial surfaces, synthesizing exopolymers in situ, which promote cell clustering and adherence to tooth enamel. We investigated the potential role Gtfs may play in mediating the interactions between C. albicans SC5314 and S. mutans UA159, both with each other and with the sHA surface. GtfB adhered effectively to the C. albicans yeast cell surface in an enzymatically active form, as determined by scintillation spectroscopy and fluorescence imaging. The glucans formed on the yeast cell surface were more susceptible to dextranase than those synthesized in solution or on sHA and bacterial cell surfaces (P < 0.05), indicating an elevated α-1,6-linked glucose content. Fluorescence imaging revealed that larger numbers of S. mutans cells bound to C. albicans cells with glucans present on their surface than to yeast cells without surface glucans (uncoated). The glucans formed in situ also enhanced C. albicans interactions with sHA, as determined by a novel single-cell micromechanical method. Furthermore, the presence of glucan-coated yeast cells significantly increased the accumulation of S. mutans on the sHA surface (versus S. mutans incubated alone or mixed with uncoated C. albicans; P < 0.05). These data reveal a novel cross-kingdom interaction that is mediated by bacterial GtfB, which readily attaches to the yeast cell surface. Surface-bound GtfB promotes the formation of a glucan-rich matrix in situ and may enhance the accumulation of S. mutans on the tooth enamel surface, thereby modulating the development of virulent biofilms.
Figures



References
-
- Banas J. A., Vickerman M. M. 2003. Glucan-binding proteins of the oral streptococci. Crit. Rev. Oral Biol. Med. 14: 89–99 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

