When gold can do what iodine cannot do: A critical comparison
- PMID: 21804881
- PMCID: PMC3135141
- DOI: 10.3762/bjoc.7.97
When gold can do what iodine cannot do: A critical comparison
Abstract
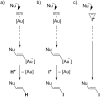
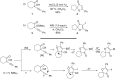
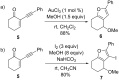
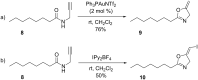
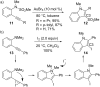
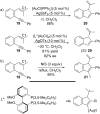
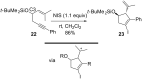
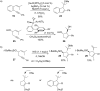
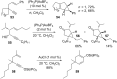
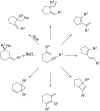
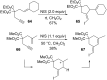
Gold catalysis has emerged as one of the most dynamic fields in organic synthesis. Only recently, more and more domino processes, for which gold pre-catalysts were found to be outstandingly effective, were paralleled by employing iodine electrophiles in place of gold compounds. This review highlights how, in certain cases, iodonium activation can match gold-catalyzed reactions to construct identical product scaffolds. Likewise, processes are discussed where mostly identical starting materials are transformed into diverse frameworks depending on whether gold or iodonium activation was used to trigger the reaction.
Keywords: catalysis; cycloisomerizations; domino reactions; gold; iodine.
Figures




Similar articles
-
Alkyne activation with Brønsted acids, iodine, or gold complexes, and its fate leading to synthetic application.Chem Commun (Camb). 2009 Sep 14;(34):5075-87. doi: 10.1039/b909978f. Epub 2009 Jul 17. Chem Commun (Camb). 2009. PMID: 20448954
-
Supported gold catalysis: from small molecule activation to green chemical synthesis.Acc Chem Res. 2014 Mar 18;47(3):793-804. doi: 10.1021/ar400165j. Epub 2013 Dec 11. Acc Chem Res. 2014. PMID: 24328524
-
Catalysis by unsupported skeletal gold catalysts.Acc Chem Res. 2014 Mar 18;47(3):731-9. doi: 10.1021/ar400202p. Epub 2013 Nov 22. Acc Chem Res. 2014. PMID: 24266888
-
Assembly of functionalized carbocycles or N-heterocycles through a domino electrocyclization-[1,2] migration reaction sequence.Org Biomol Chem. 2015 Oct 14;13(38):9720-41. doi: 10.1039/c5ob01334h. Org Biomol Chem. 2015. PMID: 26275170 Review.
-
Mechanisms in Iodine Catalysis.Chemistry. 2018 Jul 2;24(37):9187-9199. doi: 10.1002/chem.201706136. Epub 2018 Apr 30. Chemistry. 2018. PMID: 29469220 Review.
Cited by
-
Iodine-mediated synthesis of benzo[a]fluorenones from yne-enones.RSC Adv. 2019 Jul 30;9(41):23652-23657. doi: 10.1039/c9ra02376c. eCollection 2019 Jul 29. RSC Adv. 2019. PMID: 35530623 Free PMC article.
-
Synthetic approaches to multifunctional indenes.Beilstein J Org Chem. 2011;7:1739-44. doi: 10.3762/bjoc.7.204. Epub 2011 Dec 29. Beilstein J Org Chem. 2011. PMID: 22238553 Free PMC article.
-
Fluorohydration of alkynes via I(I)/I(III) catalysis.Beilstein J Org Chem. 2020 Jul 10;16:1627-1635. doi: 10.3762/bjoc.16.135. eCollection 2020. Beilstein J Org Chem. 2020. PMID: 32704329 Free PMC article.
-
Recent progress in transition-metal-free functionalization of allenamides.RSC Adv. 2020 Oct 6;10(60):36818-36827. doi: 10.1039/d0ra07119f. eCollection 2020 Oct 1. RSC Adv. 2020. PMID: 35517974 Free PMC article. Review.
References
LinkOut - more resources
Full Text Sources
