Autophosphorylation in the leucine-rich repeat kinase 2 (LRRK2) GTPase domain modifies kinase and GTP-binding activities
- PMID: 21806997
- PMCID: PMC3158845
- DOI: 10.1016/j.jmb.2011.07.033
Autophosphorylation in the leucine-rich repeat kinase 2 (LRRK2) GTPase domain modifies kinase and GTP-binding activities
Erratum in
-
Corrigendum to "Autophosphorylation in the Leucine-Rich Repeat Kinase 2 (LRRK2) GTPase Domain Modified Kinase and GTP-binding Activities" [J. Mol. Biol. 412(1) (2011) 94-110].J Mol Biol. 2021 Sep 3;433(18):167129. doi: 10.1016/j.jmb.2021.167129. Epub 2021 Jul 20. J Mol Biol. 2021. PMID: 34294467 Free PMC article. No abstract available.
Abstract
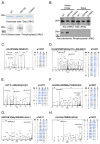
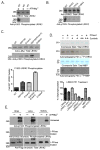
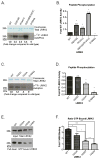
The leucine-rich repeat kinase 2 (LRRK2) protein has both guanosine triphosphatase (GTPase) and kinase activities, and mutation in either enzymatic domain can cause late-onset Parkinson disease. Nucleotide binding in the GTPase domain may be required for kinase activity, and residues in the GTPase domain are potential sites for autophosphorylation, suggesting a complex mechanism of intrinsic regulation. To further define the effects of LRRK2 autophosphorylation, we applied a technique optimal for detection of protein phosphorylation, electron transfer dissociation, and identified autophosphorylation events exclusively nearby the nucleotide binding pocket in the GTPase domain. Parkinson-disease-linked mutations alter kinase activity but did not alter autophosphorylation site specificity or sites of phosphorylation in a robust in vitro substrate myelin basic protein. Amino acid substitutions in the GTPase domain have large effects on kinase activity, as insertion of the GTPase-associated R1441C pathogenic mutation together with the G2019S kinase domain mutation resulted in a multiplicative increase (∼7-fold) in activity. Removal of a conserved autophosphorylation site (T1503) by mutation to an alanine residue resulted in greatly decreased GTP-binding and kinase activities. While autophosphorylation likely serves to potentiate kinase activity, we find that oligomerization and loss of the active dimer species occur in an ATP- and autophosphorylation-independent manner. LRRK2 autophosphorylation sites are overall robustly protected from dephosphorylation in vitro, suggesting tight control over activity in vivo. We developed highly specific antibodies targeting pT1503 but failed to detect endogenous autophosphorylation in protein derived from transgenic mice and cell lines. LRRK2 activity in vivo is unlikely to be constitutive but rather refined to specific responses.
Copyright © 2011 Elsevier Ltd. All rights reserved.
Figures



References
-
- Paisan-Ruiz C, Jain S, Evans EW, Gilks WP, Simon J, van der Brug M, Lopez de Munain A, Aparicio S, Gil AM, Khan N, Johnson J, Martinez JR, Nicholl D, Carrera IM, Pena AS, de Silva R, Lees A, Marti-Masso JF, Perez-Tur J, Wood NW, Singleton AB. Cloning of the gene containing mutations that cause PARK8-linked Parkinson’s disease. Neuron. 2004;44:595–600. - PubMed
-
- Zimprich A, Biskup S, Leitner P, Lichtner P, Farrer M, Lincoln S, Kachergus J, Hulihan M, Uitti RJ, Calne DB, Stoessl AJ, Pfeiffer RF, Patenge N, Carbajal IC, Vieregge P, Asmus F, Muller-Myhsok B, Dickson DW, Meitinger T, Strom TM, Wszolek ZK, Gasser T. Mutations in LRRK2 cause autosomal-dominant parkinsonism with pleomorphic pathology. Neuron. 2004;44:601–7. - PubMed
-
- West AB, Moore DJ, Choi C, Andrabi SA, Li X, Dikeman D, Biskup S, Zhang Z, Lim KL, Dawson VL, Dawson TM. Parkinson’s disease-associated mutations in LRRK2 link enhanced GTP-binding and kinase activities to neuronal toxicity. Hum Mol Genet. 2007;16:223–32. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

