Virological response after short-term CCR5 antagonist exposure in HIV-infected patients: frequency of subjects with virological response and associated factors
- PMID: 21807977
- PMCID: PMC3187011
- DOI: 10.1128/AAC.00753-11
Virological response after short-term CCR5 antagonist exposure in HIV-infected patients: frequency of subjects with virological response and associated factors
Abstract
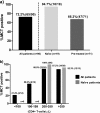
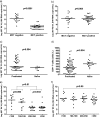
The virological response after an 8-day maraviroc monotherapy has been proposed to be an alternative method to determine whether an CCR5 antagonist should be prescribed to HIV-infected patients. The frequency of patients eligible for a combined antiretroviral therapy which includes maraviroc on the basis of the result of this clinical test is not well-known at the moment. In the same way, clinical and immunovirological factors associated with the virological response after antagonist exposure need to be determined. Ninety consecutive HIV-infected patients were exposed to an 8-day maraviroc monotherapy. The virological response was considered positive if either a reduction of ≥1-log(10) HIV RNA copies/ml or an undetectable viral load (<40 HIV RNA copies/ml) was achieved. CXCR4- and CCR5-tropic virus levels were determined by using patients' viral isolates and multiple rounds of infection of indicator cell lines (U87-CXCR4 and U87-CCR5). The frequency of patients with a positive virological response was 72.2% (94.7% and 66.2% for treatment-naïve and pretreated patients, respectively). The positive response rates dramatically decreased in patients with lower CD4(+) T-cell counts. The CXCR4-tropic virus level was the only variable independently associated with the virological response after short-term maraviroc exposure. Lower CD4(+) T-cell strata were associated with higher CXCR4-tropic virus levels. These results support the suggestion that CCR5 antagonists should be an early treatment option before the expansion of CXCR4-tropic strains.
Figures
References
-
- Berger E. A., et al. 1998. A new classification for HIV-1. Nature 391:240. - PubMed
-
- Chueca N., et al. 2009. Improvement in the determination of HIV-1 tropism using the V3 gene sequence and a combination of bioinformatic tools. J. Med. Virol. 81:763–767 - PubMed
-
- Cooper D. A., et al. 2010. Maraviroc versus efavirenz, both in combination with zidovudine-lamivudine, for the treatment of antiretroviral-naive subjects with CCR5-tropic HIV-1 infection. J. Infect. Dis. 201:803–813 - PubMed
-
- Fiser A. L., et al. 21 September 2010, posting date High CD4+ T-cell surface CXCR4 density as a risk factor for R5 to X4 switch in the course of HIV-1 infection. J. Acquir. Immune Defic. Syndr. [Epub ahead of print.] - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

