Analysis of the effect of osteon diameter on the potential relationship of osteocyte lacuna density and osteon wall thickness
- PMID: 21809466
- PMCID: PMC3277654
- DOI: 10.1002/ar.21452
Analysis of the effect of osteon diameter on the potential relationship of osteocyte lacuna density and osteon wall thickness
Abstract
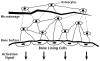
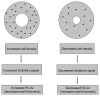
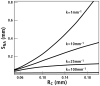
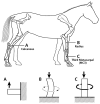
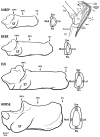
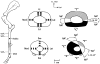
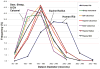
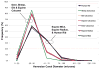
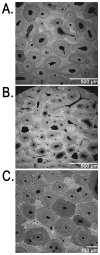
An important hypothesis is that the degree of infilling of secondary osteons (Haversian systems) is controlled by the inhibitory effect of osteocytes on osteoblasts, which might be mediated by sclerostin (a glycoprotein produced by osteocytes). Consequently, this inhibition could be proportional to cell number: relatively greater repression is exerted by progressively greater osteocyte density (increased osteocytes correlate with thinner osteon walls). This hypothesis has been examined, but only weakly supported, in sheep ulnae. We looked for this inverse relationship between osteon wall thickness (On.W.Th) and osteocyte lacuna density (Ot.Lc.N/B.Ar) in small and large osteons in human ribs, calcanei of sheep, deer, elk, and horses, and radii and third metacarpals of horses. Analyses involved: (1) all osteons, (2) smaller osteons, either ≤150 μm diameter or less than or equal to the mean diameter, and (3) larger osteons (>mean diameter). Significant, but weak, correlations between Ot.Lc.N/B.Ar and On.W.Th/On.Dm (On.Dm = osteon diameter) were found when considering all osteons in limb bones (r values -0.16 to -0.40, P < 0.01; resembling previous results in sheep ulnae: r = -0.39, P < 0.0001). In larger osteons, these relationships were either not significant (five/seven bone types) or very weak (two/seven bone types). In ribs, a negative relationship was only found in smaller osteons (r = -0.228, P < 0.01); this inverse relationship in smaller osteons did not occur in elk calcanei. These results do not provide clear or consistent support for the hypothesized inverse relationship. However, correlation analyses may fail to detect osteocyte-based repression of infilling if the signal is spatially nonuniform (e.g., increased near the central canal).
Keywords: bone remodeling; osteocytes; osteon wall thickness; osteons; sclerostin.
Copyright © 2011 Wiley-Liss, Inc.
Figures

Similar articles
-
Morphometric analysis of osteonal architecture in bones from healthy young human male subjects using scanning electron microscopy.J Anat. 2013 Sep;223(3):242-54. doi: 10.1111/joa.12079. Epub 2013 Jul 8. J Anat. 2013. PMID: 23834434 Free PMC article.
-
Histomorphometric assessment of Haversian canal and osteocyte lacunae in different-sized osteons in human rib.Anat Rec A Discov Mol Cell Evol Biol. 2003 Jun;272(2):520-5. doi: 10.1002/ar.a.10058. Anat Rec A Discov Mol Cell Evol Biol. 2003. PMID: 12740946
-
Secondary osteon size and collagen/lamellar organization ("osteon morphotypes") are not coupled, but potentially adapt independently for local strain mode or magnitude.J Struct Biol. 2013 Feb;181(2):95-107. doi: 10.1016/j.jsb.2012.10.013. Epub 2012 Nov 1. J Struct Biol. 2013. PMID: 23123271
-
Osteon: Structure, Turnover, and Regeneration.Tissue Eng Part B Rev. 2022 Apr;28(2):261-278. doi: 10.1089/ten.TEB.2020.0322. Epub 2021 Mar 8. Tissue Eng Part B Rev. 2022. PMID: 33487116 Free PMC article. Review.
-
The actions of parathyroid hormone on bone: relation to bone remodeling and turnover, calcium homeostasis, and metabolic bone disease. Part I of IV parts: mechanisms of calcium transfer between blood and bone and their cellular basis: morphological and kinetic approaches to bone turnover.Metabolism. 1976 Jul;25(7):809-44. doi: 10.1016/0026-0495(76)90151-7. Metabolism. 1976. PMID: 781470 Review.
Cited by
-
Secondary osteons scale allometrically in mammalian humerus and femur.R Soc Open Sci. 2017 Nov 8;4(11):170431. doi: 10.1098/rsos.170431. eCollection 2017 Nov. R Soc Open Sci. 2017. PMID: 29291052 Free PMC article.
-
Morphometric analysis of osteonal architecture in bones from healthy young human male subjects using scanning electron microscopy.J Anat. 2013 Sep;223(3):242-54. doi: 10.1111/joa.12079. Epub 2013 Jul 8. J Anat. 2013. PMID: 23834434 Free PMC article.
-
The effect of caponization on tibia bone histomorphometric properties of crossbred roosters.Sci Rep. 2024 Feb 19;14(1):4062. doi: 10.1038/s41598-024-54791-6. Sci Rep. 2024. PMID: 38374163 Free PMC article.
-
Bone Abrasive Machining: Influence of Tool Geometry and Cortical Bone Anisotropic Structure on Crack Propagation.J Funct Biomater. 2022 Sep 15;13(3):154. doi: 10.3390/jfb13030154. J Funct Biomater. 2022. PMID: 36135588 Free PMC article.
-
Normal variation in cortical osteocyte lacunar parameters in healthy young males.J Anat. 2014 Sep;225(3):328-36. doi: 10.1111/joa.12213. Epub 2014 Jul 4. J Anat. 2014. PMID: 25040136 Free PMC article.
References
-
- Aguirre JI, Plotkin LI, Stewart SA, Weinstein RS, Parfitt AM, Manolagas SC, Bellido T. Osteocyte apoptosis is induced by weightlessness in mice and precedes osteoclast recruitment and bone loss. J Bone Miner Res. 2006;21:605–615. - PubMed
-
- Bachus KN, Bloebaum RD. Projection effect errors in biomaterials and bone research. Cells Mater. 1992;2:347–355.
-
- Bellido T, Ali AA, Gubrij I, Plotkin LI, Fu Q, O’Brien CA, Manolagas SC, Jilka RL. Chronic elevation of parathyroid hormone in mice reduces expression of sclerostin by osteocytes: a novel mechanism for hormonal control of osteoblastogenesis. Endocrinology. 2005;146:4577–4583. - PubMed
-
- Biewener AA, Thomason J, Goodship A, Lanyon LE. Bone stress in the horse forelimb during locomotion at different gaits: A comparison of two experimental methods. J Biomech. 1983a;16:565–576. - PubMed
-
- Biewener AA, Thomason J, Lanyon LE. Mechanics of locomotion and jumping in the forelimb of the horse (Equus): In vivo stress developed in the radius and metacarpus. J Zool Lond. 1983b;201:67–82.
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

