Detailed protocol for evaluation of dynamic perifusion of human islets to assess β-cell function
- PMID: 21811103
- PMCID: PMC3219161
- DOI: 10.4161/isl.3.5.15938
Detailed protocol for evaluation of dynamic perifusion of human islets to assess β-cell function
Abstract
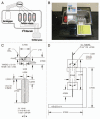
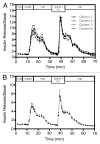
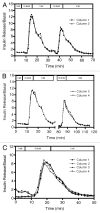
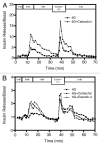
The definitive measure of β-cell quality in an islet is the measurement of β-cell function, i.e., the ability of the islets to release insulin in a controlled manner in response to minute changes in ambient glucose levels. Continuous flow or dynamic perifusion of the solution containing glucose and secretagogues through the islets is the most accurate assessment of regulated insulin release in vitro. Here, we describe in detail a low cost, mini-perifusion system that can be adapted to any laboratory to assess islet function by examining dynamic insulin release in response to elevated glucose concentrations and addition of secretagogues. Human islets with purity > 80% and viability > 90% were perifused with low glucose (1 mM) and subsequently challenged with high glucose (16.8 mM ± KCl, 25 mM). A prototypical biphasic response to elevated glucose concentrations was observed with an average 8-fold (above basal) increase in insulin concentration at peak values. Similarly, perifusion with carbachol or exendin-4 (Byetta) with glucose (6 mM) resulted in 1.32- and 1.35-fold increase in insulin secretion above basal. Islets could be maintained in the perifusion apparatus and continued to respond to glucose for up to 3 h. At minimal financial cost and technical expertise, this apparatus can be set-up in any biological laboratory to evaluate regulated hormone release from many cell types in less than 6 h. This will allow other laboratories to measure insulin responses to their drug or modifier of interest in vitro, in a manner that better approximates islet function in vivo.
Figures
References
-
- Ricordi C, Lacy PE, Scharp DW. Automated islet isolation from human pancreas. Diabetes. 1989;38:140–142. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
