Significance of heparanase in cancer and inflammation
- PMID: 21811836
- PMCID: PMC3399068
- DOI: 10.1007/s12307-011-0082-7
Significance of heparanase in cancer and inflammation
Abstract
Heparan sulfate proteoglycans (HSPGs) are primary components at the interface between virtually every eukaryotic cell and its extracellular matrix. HSPGs not only provide a storage depot for heparin-binding molecules in the cell microenvironment, but also decisively regulate their accessibility, function and mode of action. As such, they are intimately involved in modulating cell invasion and signaling loops that are critical for tumor growth, inflammation and kidney function. In a series of studies performed since the cloning of the human heparanase gene, we and others have demonstrated that heparanase, the sole heparan sulfate degrading endoglycosidase, is causally involved in cancer progression, inflammation and diabetic nephropathy and hence is a valid target for drug development. Heparanase is causally involved in inflammation and accelerates colon tumorigenesis associated with inflammatory bowel disease. Notably, heparanase stimulates macrophage activation, while macrophages induce production and activation of latent heparanase contributed by the colon epithelium, together generating a vicious cycle that powers colitis and the associated tumorigenesis. Heparanase also plays a decisive role in the pathogenesis of diabetic nephropathy, degrading heparan sulfate in the glomerular basement membrane and ultimately leading to proteinuria and kidney dysfunction. Notably, clinically relevant doses of ionizing radiation (IR) upregulate heparanase expression and thereby augment the metastatic potential of pancreatic carcinoma. Thus, combining radiotherapy with heparanase inhibition is an effective strategy to prevent tumor resistance and dissemination in IR-treated pancreatic cancer patients. Also, accumulating evidence indicate that peptides derived from human heparanase elicit a potent anti-tumor immune response, suggesting that heparanase represents a promising target antigen for immunotherapeutic approaches against a broad variety of tumours. Oligosaccharide-based compounds that inhibit heparanase enzymatic activity were developed, aiming primarily at halting tumor growth, metastasis and angiogenesis. Some of these compounds are being evaluated in clinical trials, targeting both the tumor and tumor microenvironment.
Figures


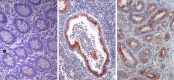

References
-
- Vlodavsky I, Gospodarowicz D. Respective roles of laminin and fibronectin in adhesion of human carcinoma and sarcoma cells. Nature. 1981;289:304–306. - PubMed
-
- Vlodavsky I, Lui GM, Gospodarowicz D. Morphological appearance, growth behavior and migratory activity of human tumor cells maintained on extracellular matrix versus plastic. Cell. 1980;19:607–616. - PubMed
-
- Vlodavsky I (2001) Preparation of extracellular matrices produced by cultured corneal endothelial and PF-HR9 endodermal cells. Curr Protoc Cell Biol. Chapter 10:Unit 10.4 - PubMed
-
- Kleinman HK, Martin GR. Matrigel: basement membrane matrix with biological activity. Semin Cancer Biol. 2005;15:378–386. - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

