Physiologic control of IDO competence in splenic dendritic cells
- PMID: 21813777
- PMCID: PMC3556270
- DOI: 10.4049/jimmunol.1100276
Physiologic control of IDO competence in splenic dendritic cells
Abstract
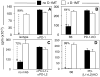
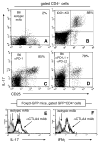
Dendritic cells (DCs) competent to express the regulatory enzyme IDO in mice are a small but distinctive subset of DCs. Previously, we reported that a high-dose systemic CpG treatment to ligate TLR9 in vivo induced functional IDO exclusively in splenic CD19(+) DCs, which stimulated resting Foxp3-lineage regulatory T cells (Tregs) to rapidly acquire potent suppressor activity. In this paper, we show that IDO was induced in spleen and peripheral lymph nodes after CpG treatment in a dose-dependent manner. Induced IDO suppressed local T cell responses to exogenous Ags and inhibited proinflammatory cytokine expression in response to TLR9 ligation. IDO induction did not occur in T cell-deficient mice or in mice with defective B7 or programmed death (PD)-1 costimulatory pathways. Consistent with these findings, CTLA4 or PD-1/PD-ligand costimulatory blockade abrogated IDO induction and prevented Treg activation via IDO following high-dose CpG treatment. Consequently, CD4(+)CD25(+) T cells uniformly expressed IL-17 shortly after TLR9 ligation. These data support the hypothesis that constitutive interactions from activated T cells or Tregs and IDO-competent DCs via concomitant CTLA4→B7 and PD-1→PD-ligand signals maintain the default potential to regulate T cell responsiveness via IDO. Acute disruption of these nonredundant interactions abrogated regulation via IDO, providing novel perspectives on the proinflammatory effects of costimulatory blockade therapies. Moreover, interactions between IDO-competent DCs and activated T cells in lymphoid tissues may attenuate proinflammatory responses to adjuvants such as TLR ligands.
Figures




References
-
- Cobbold SP, Adams E, Nolan KF, Regateiro FS, Waldmann H. Connecting the mechanisms of T-cell regulation: dendritic cells as the missing link. Immunol Rev. 2010;236:203–218. - PubMed
-
- Mellor AL, Munn DH. Creating immune privilege: active local suppression that benefits friends, but protects foes. Nat Rev Immunol. 2008;8:74–80. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
- P01 AI056299/AI/NIAID NIH HHS/United States
- R01 HD041187/HD/NICHD NIH HHS/United States
- P01 CA065493/CA/NCI NIH HHS/United States
- CA096651/CA/NCI NIH HHS/United States
- R37 AI034495/AI/NIAID NIH HHS/United States
- R01 CA103320/CA/NCI NIH HHS/United States
- R01 CA096651/CA/NCI NIH HHS/United States
- AI34495/AI/NIAID NIH HHS/United States
- R01 AI034495/AI/NIAID NIH HHS/United States
- P01 AI 56299/AI/NIAID NIH HHS/United States
- CA72669/CA/NCI NIH HHS/United States
- R01 CA072669/CA/NCI NIH HHS/United States
- P01 CA142106/CA/NCI NIH HHS/United States
- CA103320/CA/NCI NIH HHS/United States
- R01 AI044219/AI/NIAID NIH HHS/United States
- AI63402/AI/NIAID NIH HHS/United States
- AI75165/AI/NIAID NIH HHS/United States
- R37 HL056067/HL/NHLBI NIH HHS/United States
- R01 HL056067/HL/NHLBI NIH HHS/United States
- AI056299/AI/NIAID NIH HHS/United States
- R01 AI063402/AI/NIAID NIH HHS/United States
- HL56067/HL/NHLBI NIH HHS/United States
- R01 CA112431/CA/NCI NIH HHS/United States
- R01 AI075165/AI/NIAID NIH HHS/United States
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

