A prion-like trigger of antiviral signaling
- PMID: 21816270
- PMCID: PMC4272580
- DOI: 10.1016/j.cell.2011.07.018
A prion-like trigger of antiviral signaling
Abstract
The MAVS protein plays a critical role in the assembly of an antiviral signaling complex on mitochondrial membranes. Hou et al. (2011) now report that virus infection induces a conformational change in MAVS, leading to the prion-like formation of functional self-aggregates that provide a sensitive trigger for antiviral signaling.
Copyright © 2011 Elsevier Inc. All rights reserved.
Figures
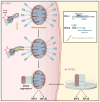
Comment on
-
MAVS forms functional prion-like aggregates to activate and propagate antiviral innate immune response.Cell. 2011 Aug 5;146(3):448-61. doi: 10.1016/j.cell.2011.06.041. Epub 2011 Jul 21. Cell. 2011. PMID: 21782231 Free PMC article.
References
-
- Seth RB, Sun L, Ea CK, Chen ZJ. Cell. 2005;122:669–682. - PubMed
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

