Activation of mitogen-activated protein kinases by 5,6-dimethylxanthenone-4-acetic acid (DMXAA) plays an important role in macrophage stimulation
- PMID: 21819972
- PMCID: PMC3191304
- DOI: 10.1016/j.bcp.2011.07.086
Activation of mitogen-activated protein kinases by 5,6-dimethylxanthenone-4-acetic acid (DMXAA) plays an important role in macrophage stimulation
Abstract
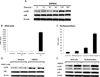
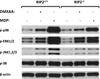
The small molecule anti-tumor agent, 5,6-dimethylxanthenone-4-acetic acid (DMXAA, now called Vadimezan) is a potent macrophage and dendritic cell activating agent that, in the murine system, results in the release of large amounts of cytokines and chemokines. The mechanisms by which this release is mediated have not been fully elucidated. The mitogen-activated protein kinase (MAPK) pathways play an important role in the regulation of proinflammatory cytokines, such as TNF-α, IL-1β, as well as the responses to extracellular stimuli, such as lipopolysaccharide (LPS). The results of this study demonstrate that DMXAA activates three members of mitogen-activated protein kinase (MAPK) superfamily, namely p38 MAPK, extracellular signal-regulated kinases 1 and 2 (ERK1 and ERK2), and c-Jun N-terminal kinases (JNKs) via a RIP2-independent mechanism in murine macrophages. By using selective inhibitors of MAPKs, this study confirms that both activated p38/MK2 pathways and ERK1/2 MAPK play a significant role in regulation of both TNF-α and IL-6 protein production induced by DMXAA at the post-transcriptional level. Our findings also show that interferon-γ priming can dramatically augment TNF-α protein secretion induced by DMXAA through enhancing activation of multiple MAPK pathways at the post-transcriptional level. This study expands current knowledge on mechanisms of how DMXAA acts as a potent anti-tumor agent in murine system and also provides useful information for further study on the mechanism of action of this potential anti-tumor compound in human macrophages.
Copyright © 2011 Elsevier Inc. All rights reserved.
Figures




Similar articles
-
p38 mitogen-activated protein kinase is required for the antitumor activity of the vascular disrupting agent 5,6-dimethylxanthenone-4-acetic acid.J Pharmacol Exp Ther. 2012 Jun;341(3):709-17. doi: 10.1124/jpet.112.191635. Epub 2012 Mar 13. J Pharmacol Exp Ther. 2012. PMID: 22414857
-
Mitogen-activated protein kinases and nuclear factor-kappaB regulate Helicobacter pylori-mediated interleukin-8 release from macrophages.Biochem J. 2002 Nov 15;368(Pt 1):121-9. doi: 10.1042/BJ20020555. Biochem J. 2002. PMID: 12150710 Free PMC article.
-
Activation of tumor-associated macrophages by the vascular disrupting agent 5,6-dimethylxanthenone-4-acetic acid induces an effective CD8+ T-cell-mediated antitumor immune response in murine models of lung cancer and mesothelioma.Cancer Res. 2005 Dec 15;65(24):11752-61. doi: 10.1158/0008-5472.CAN-05-1658. Cancer Res. 2005. PMID: 16357188
-
Transcriptional and posttranscriptional roles for p38 mitogen-activated protein kinase in upregulation of TNF-alpha expression by deoxynivalenol (vomitoxin).Toxicol Appl Pharmacol. 2003 Dec 1;193(2):188-201. doi: 10.1016/s0041-008x(03)00299-0. Toxicol Appl Pharmacol. 2003. PMID: 14644621
-
An overview on Vadimezan (DMXAA): The vascular disrupting agent.Chem Biol Drug Des. 2018 May;91(5):996-1006. doi: 10.1111/cbdd.13166. Epub 2018 Jan 24. Chem Biol Drug Des. 2018. PMID: 29288534 Review.
Cited by
-
Myeloid Cells as Clinical Biomarkers for Immune Checkpoint Blockade.Front Immunol. 2020 Jul 24;11:1590. doi: 10.3389/fimmu.2020.01590. eCollection 2020. Front Immunol. 2020. PMID: 32793228 Free PMC article. Review.
-
5,6-Dimethylxanthenone-4-acetic acid (DMXAA) activates stimulator of interferon gene (STING)-dependent innate immune pathways and is regulated by mitochondrial membrane potential.J Biol Chem. 2012 Nov 16;287(47):39776-88. doi: 10.1074/jbc.M112.382986. Epub 2012 Oct 1. J Biol Chem. 2012. PMID: 23027866 Free PMC article.
-
The chemotherapeutic agent DMXAA as a unique IRF3-dependent type-2 vaccine adjuvant.PLoS One. 2013;8(3):e60038. doi: 10.1371/journal.pone.0060038. Epub 2013 Mar 21. PLoS One. 2013. PMID: 23555875 Free PMC article.
-
Pharmacologic activation of the innate immune system to prevent respiratory viral infections.Am J Respir Cell Mol Biol. 2011 Sep;45(3):480-8. doi: 10.1165/rcmb.2010-0288OC. Epub 2010 Dec 10. Am J Respir Cell Mol Biol. 2011. PMID: 21148741 Free PMC article.
-
Identification of human-selective analogues of the vascular-disrupting agent 5,6-dimethylxanthenone-4-acetic acid (DMXAA).Br J Cancer. 2013 Apr 2;108(6):1306-15. doi: 10.1038/bjc.2013.101. Epub 2013 Mar 12. Br J Cancer. 2013. PMID: 23481185 Free PMC article.
References
-
- Ching LM, Goldsmith D, Joseph WR, Korner H, Sedgwick JD, Baguley BC. Induction of intratumoral tumor necrosis factor (TNF) synthesis and hemorrhagic necrosis by 5,6-dimethylxanthenone-4-acetic acid (DMXAA) in TNF knockout mice. Cancer Res. 1999;59:3304–3307. - PubMed
-
- Jassar AS, Suzuki E, Kapoor V, Sun J, Silverberg MB, Cheung L, et al. Activation of tumor-associated macrophages by the vascular disrupting agent 5,6-dimethylxanthenone-4-acetic acid induces an effective CD8+ T-cell-mediated antitumor immune response in murine models of lung cancer and mesothelioma. Cancer Res. 2005;65:11752–11761. - PubMed
-
- Wallace A, LaRosa DF, Kapoor V, Sun J, Cheng G, Jassar A, et al. The vascular disrupting agent, DMXAA, directly activates dendritic cells through a MyD88-independent mechanism and generates antitumor cytotoxic T lymphocytes. Cancer Res. 2007;67:7011–7019. - PubMed
-
- Ching LM, Young HA, Eberly K, Yu CR. Induction of STAT and NFkappaB activation by the antitumor agents 5,6-dimethylxanthenone-4-acetic acid and flavone acetic acid in a murine macrophage cell line. Biochem Pharmacol. 1999;58:1173–1181. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

