Two-dimensional infrared spectroscopy of azido-nicotinamide adenine dinucleotide in water
- PMID: 21823737
- PMCID: PMC3162616
- DOI: 10.1063/1.3623418
Two-dimensional infrared spectroscopy of azido-nicotinamide adenine dinucleotide in water
Abstract
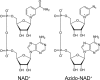
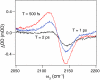
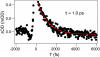
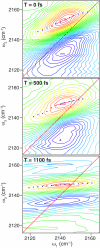
Mid-IR active analogs of enzyme cofactors have the potential to be important spectroscopic reporters of enzyme active site dynamics. Azido-nicotinamide adenine dinucleotide (NAD(+)), which has been recently synthesized in our laboratory, is a mid-IR active analog of NAD(+), a ubiquitous redox cofactor in biology. In this study, we measure the frequency-frequency time correlation function for the antisymmetric stretching vibration of the azido group of azido-NAD(+) in water. Our results are consistent with previous studies of pseudohalides in water. We conclude that azido-NAD(+) is sensitive to local environmental fluctuations, which, in water, are dominated by hydrogen-bond dynamics of the water molecules around the probe. Our results demonstrate the potential of azido-NAD(+) as a vibrational probe and illustrate the potential of substituted NAD(+)-analogs as reporters of local structural dynamics that could be used for studies of protein dynamics in NAD-dependent enzymes.
Figures
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

