Evolution of functional nucleic acids in the presence of nonheritable backbone heterogeneity
- PMID: 21825162
- PMCID: PMC3158193
- DOI: 10.1073/pnas.1107113108
Evolution of functional nucleic acids in the presence of nonheritable backbone heterogeneity
Abstract
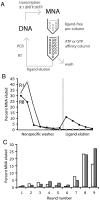
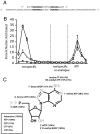
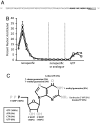
Multiple lines of evidence support the hypothesis that the early evolution of life was dominated by RNA, which can both transfer information from generation to generation through replication directed by base-pairing, and carry out biochemical activities by folding into functional structures. To understand how life emerged from prebiotic chemistry we must therefore explain the steps that led to the emergence of the RNA world, and in particular, the synthesis of RNA. The generation of pools of highly pure ribonucleotides on the early Earth seems unlikely, but the presence of alternative nucleotides would support the assembly of nucleic acid polymers containing nonheritable backbone heterogeneity. We suggest that homogeneous monomers might not have been necessary if populations of heterogeneous nucleic acid molecules could evolve reproducible function. For such evolution to be possible, function would have to be maintained despite the repeated scrambling of backbone chemistry from generation to generation. We have tested this possibility in a simplified model system, by using a T7 RNA polymerase variant capable of transcribing nucleic acids that contain an approximately 11 mixture of deoxy- and ribonucleotides. We readily isolated nucleotide-binding aptamers by utilizing an in vitro selection process that shuffles the order of deoxy- and ribonucleotides in each round. We describe two such RNA/DNA mosaic nucleic acid aptamers that specifically bind ATP and GTP, respectively. We conclude that nonheritable variations in nucleic acid backbone structure may not have posed an insurmountable barrier to the emergence of functionality in early nucleic acids.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
Similar articles
-
The Emergence of RNA from the Heterogeneous Products of Prebiotic Nucleotide Synthesis.J Am Chem Soc. 2021 Mar 10;143(9):3267-3279. doi: 10.1021/jacs.0c12955. Epub 2021 Feb 26. J Am Chem Soc. 2021. PMID: 33636080 Review.
-
Primitive genetic polymers.Cold Spring Harb Perspect Biol. 2010 Dec;2(12):a002196. doi: 10.1101/cshperspect.a002196. Epub 2010 May 12. Cold Spring Harb Perspect Biol. 2010. PMID: 20462999 Free PMC article. Review.
-
Experimental evidence that GNA and TNA were not sequential polymers in the prebiotic evolution of RNA.J Mol Evol. 2007 Sep;65(3):289-95. doi: 10.1007/s00239-007-9017-9. Epub 2007 Sep 9. J Mol Evol. 2007. PMID: 17828568
-
A Nucleic Acid Sequence That is Catalytically Active in Both RNA and TNA Backbones.ACS Synth Biol. 2022 Nov 18;11(11):3874-3885. doi: 10.1021/acssynbio.2c00479. Epub 2022 Oct 24. ACS Synth Biol. 2022. PMID: 36278399
-
Chemical etiology of nucleic acid structure: the pentulofuranosyl oligonucleotide systems: the (1'→3')-β-L-ribulo, (4'→3')-α-L-xylulo, and (1'→3')-α-L-xylulo nucleic acids.Chemistry. 2013 Nov 4;19(45):15336-45. doi: 10.1002/chem.201302219. Epub 2013 Sep 23. Chemistry. 2013. PMID: 24150882
Cited by
-
Emergence of native peptide sequences in prebiotic replication networks.Nat Commun. 2017 Sep 5;8(1):434. doi: 10.1038/s41467-017-00463-1. Nat Commun. 2017. PMID: 28874657 Free PMC article.
-
Impact of template backbone heterogeneity on RNA polymerase II transcription.Nucleic Acids Res. 2015 Feb 27;43(4):2232-41. doi: 10.1093/nar/gkv059. Epub 2015 Feb 6. Nucleic Acids Res. 2015. PMID: 25662224 Free PMC article.
-
The free energy landscape of pseudorotation in 3'-5' and 2'-5' linked nucleic acids.J Am Chem Soc. 2014 Feb 19;136(7):2858-65. doi: 10.1021/ja412079b. Epub 2014 Feb 5. J Am Chem Soc. 2014. PMID: 24499340 Free PMC article.
-
Elucidating the molecular architecture of adaptation via evolve and resequence experiments.Nat Rev Genet. 2015 Oct;16(10):567-82. doi: 10.1038/nrg3937. Epub 2015 Sep 8. Nat Rev Genet. 2015. PMID: 26347030 Free PMC article. Review.
-
A Model for the Emergence of RNA from a Prebiotically Plausible Mixture of Ribonucleotides, Arabinonucleotides, and 2'-Deoxynucleotides.J Am Chem Soc. 2020 Feb 5;142(5):2317-2326. doi: 10.1021/jacs.9b11239. Epub 2020 Jan 22. J Am Chem Soc. 2020. PMID: 31913615 Free PMC article.
References
-
- Joyce GF. RNA evolution and the origins of life. Nature. 1989;338:217–224. - PubMed
-
- Piccirilli JA. Origin of life. RNA seeks its maker. Nature. 1995;376:548–549. - PubMed
-
- Engelhart AE, Hud NV. Primitive genetic polymers. Cold Spring Harb Perspect Biol. 2010;2 doi: 10.1101/cshperspect.a002196. - DOI - PMC - PubMed
-
- Eschenmoser A. Chemical etiology of nucleic acid structure. Science. 1999;284:2118–2124. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

