Gelsolin and non-muscle myosin IIA interact to mediate calcium-regulated collagen phagocytosis
- PMID: 21828045
- PMCID: PMC3190781
- DOI: 10.1074/jbc.M111.247783
Gelsolin and non-muscle myosin IIA interact to mediate calcium-regulated collagen phagocytosis
Abstract
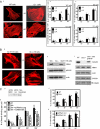
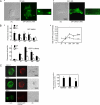
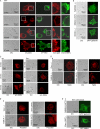
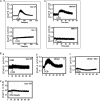
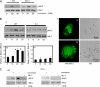
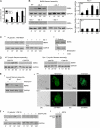
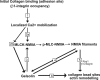
The formation of adhesion complexes is the rate-limiting step for collagen phagocytosis by fibroblasts, but the role of Ca(2+) and the potential interactions of actin-binding proteins in regulating collagen phagocytosis are not well defined. We found that the binding of collagen beads to fibroblasts was temporally and spatially associated with actin assembly at nascent phagosomes, which was absent in gelsolin null cells. Analysis of tryptic digests isolated from gelsolin immunoprecipitates indicated that non-muscle (NM) myosin IIA may bind to gelsolin. Immunostaining and immunoprecipitation showed that gelsolin and NM myosin IIA associated at collagen adhesion sites. Gelsolin and NM myosin IIA were both required for collagen binding and internalization. Collagen binding to cells initiated a prolonged increase of [Ca(2+)](i), which was absent in cells null for gelsolin or NM myosin IIA. Collagen bead-induced increases of [Ca(2+)](i) were associated with phosphorylation of the myosin light chain, which was dependent on gelsolin. NM myosin IIA filament assembly, which was dependent on myosin light chain phosphorylation and increased [Ca(2+)](i), also required gelsolin. Ionomycin-induced increases of [Ca(2+)](i) overcame the block of myosin filament assembly in gelsolin null cells. We conclude that gelsolin and NM myosin IIA interact at collagen adhesion sites to enable NM myosin IIA filament assembly and localized, Ca(2+)-dependent remodeling of actin at the nascent phagosome and that these steps are required for collagen phagocytosis.
Figures

References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

