The evolution of nervous system patterning: insights from sea urchin development
- PMID: 21828090
- PMCID: PMC3152920
- DOI: 10.1242/dev.058172
The evolution of nervous system patterning: insights from sea urchin development
Abstract
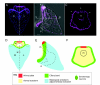
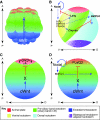
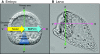
Recent studies of the sea urchin embryo have elucidated the mechanisms that localize and pattern its nervous system. These studies have revealed the presence of two overlapping regions of neurogenic potential at the beginning of embryogenesis, each of which becomes progressively restricted by separate, yet linked, signals, including Wnt and subsequently Nodal and BMP. These signals act to specify and localize the embryonic neural fields - the anterior neuroectoderm and the more posterior ciliary band neuroectoderm - during development. Here, we review these conserved nervous system patterning signals and consider how the relationships between them might have changed during deuterostome evolution.
Figures


References
-
- Angerer L. M., Oleksyn D. W., Logan C. Y., McClay D. R., Dale L., Angerer R. C. (2000). A BMP pathway regulates cell fate allocation along the sea urchin animal-vegetal embryonic axis. Development 127, 1105-1114 - PubMed
-
- Angerer L. M., Oleksyn D. W., Levine A. M., Li X., Klein W. H., Angerer R. C. (2001). Sea urchin goosecoid function links fate specification along the animal-vegetal and oral-aboral embryonic axes. Development 128, 4393-4404 - PubMed
-
- Bergeron K. F., Xu X., Brandhorst B. P. (2010). Oral-aboral patterning and gastrulation of sea urchin embryos depend on sulfated glycosaminoglycans. Mech. Dev. 128, 71-89 - PubMed
-
- Bisgrove B. W., Burke R. D. (1986). Development of serotongic neurons in embryos of the sea urchin, Strongylocentrotus purpuratus. Dev. Growth Differ. 28, 569-574 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

