Tumor-derived exosomes confer antigen-specific immunosuppression in a murine delayed-type hypersensitivity model
- PMID: 21829629
- PMCID: PMC3149056
- DOI: 10.1371/journal.pone.0022517
Tumor-derived exosomes confer antigen-specific immunosuppression in a murine delayed-type hypersensitivity model
Abstract
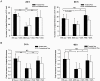
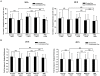
Exosomes are endosome-derived small membrane vesicles that are secreted by most cell types including tumor cells. Tumor-derived exosomes usually contain tumor antigens and have been used as a source of tumor antigens to stimulate anti-tumor immune responses. However, many reports also suggest that tumor-derived exosomes can facilitate tumor immune evasion through different mechanisms, most of which are antigen-independent. In the present study we used a mouse model of delayed-type hypersensitivity (DTH) and demonstrated that local administration of tumor-derived exosomes carrying the model antigen chicken ovalbumin (OVA) resulted in the suppression of DTH response in an antigen-specific manner. Analysis of exosome trafficking demonstrated that following local injection, tumor-derived exosomes were internalized by CD11c+ cells and transported to the draining LN. Exosome-mediated DTH suppression is associated with increased mRNA levels of TGF-β1 and IL-4 in the draining LN. The tumor-derived exosomes examined were also found to inhibit DC maturation. Taken together, our results suggest a role for tumor-derived exosomes in inducing tumor antigen-specific immunosuppression, possibly by modulating the function of APCs.
Conflict of interest statement
Figures





References
-
- Pardoll D. Does the immune system see tumors as foreign or self? Annu Rev Immunol. 2003;21:807–839. - PubMed
-
- Zou W. Immunosuppressive networks in the tumour environment and their therapeutic relevance. Nat Rev Cancer. 2005;5:263–274. - PubMed
-
- Marigo I, Dolcetti L, Serafini P, Zanovello P, Bronte V. Tumor-induced tolerance and immune suppression by myeloid derived suppressor cells. Immunol Rev. 2008;222:162–179. - PubMed
-
- Marincola FM, Jaffee EM, Hicklin DJ, Ferrone S. Escape of human solid tumors from T-cell recognition: molecular mechanisms and functional significance. Adv Immunol. 2000;74:181–273. - PubMed
-
- Thery C, Ostrowski M, Segura E. Membrane vesicles as conveyors of immune responses. Nat Rev Immunol. 2009;9:581–593. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

