Workflow and atlas system for brain-wide mapping of axonal connectivity in rat
- PMID: 21829640
- PMCID: PMC3148247
- DOI: 10.1371/journal.pone.0022669
Workflow and atlas system for brain-wide mapping of axonal connectivity in rat
Abstract
Detailed knowledge about the anatomical organization of axonal connections is important for understanding normal functions of brain systems and disease-related dysfunctions. Such connectivity data are typically generated in neuroanatomical tract-tracing experiments in which specific axonal connections are visualized in histological sections. Since journal publications typically only accommodate restricted data descriptions and example images, literature search is a cumbersome way to retrieve overviews of brain connectivity. To explore more efficient ways of mapping, analyzing, and sharing detailed axonal connectivity data from the rodent brain, we have implemented a workflow for data production and developed an atlas system tailored for online presentation of axonal tracing data. The system is available online through the Rodent Brain WorkBench (www.rbwb.org; Whole Brain Connectivity Atlas) and holds experimental metadata and high-resolution images of histological sections from experiments in which axonal tracers were injected in the primary somatosensory cortex. We here present the workflow and the data system, and exemplify how the online image repository can be used to map different aspects of the brain-wide connectivity of the rat primary somatosensory cortex, including not only presence of connections but also morphology, densities, and spatial organization. The accuracy of the approach is validated by comparing results generated with our system with findings reported in previous publications. The present study is a contribution to a systematic mapping of rodent brain connections and represents a starting point for further large-scale mapping efforts.
Conflict of interest statement
Figures
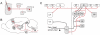
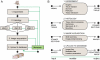

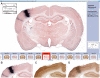
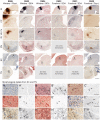

References
-
- Albin RL, Young AB, Penney JB. The functional anatomy of basal ganglia disorders. Trends Neurosci. 1989;12:366–375. - PubMed
-
- Houk JC, Wise SP. Distributed modular architectures linking basal ganglia, cerebellum, and cerebral cortex: their role in planning and controlling action. Cereb Cortex. 1995;5:95–110. - PubMed
-
- Alexander GE, Crutcher MD, DeLong MR. Basal ganglia-thalamocortical circuits: parallel substrates for motor, oculomotor, “prefrontal” and “limbic” functions. Prog Brain Res. 1990;85:119–146. - PubMed
-
- Tekin S, Cummings JL. Frontal-subcortical neuronal circuits and clinical neuropsychiatry: an update. J Psychosom Res. 2002;53:647–654. - PubMed
-
- Saleem KS, Pauls JM, Augath M, Trinath T, Prause BA, et al. Magnetic resonance imaging of neuronal connections in the macaque monkey. Neuron. 2002;34:685–700. - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources

