West Nile virus infection induces depletion of IFNAR1 protein levels
- PMID: 21830897
- PMCID: PMC3154464
- DOI: 10.1089/vim.2010.0126
West Nile virus infection induces depletion of IFNAR1 protein levels
Abstract
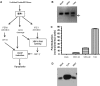
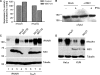
Productive virus infection requires evasion, inhibition, or subversion of innate immune responses. West Nile virus (WNV), a human pathogen that can cause symptomatic infections associated with meningitis and encephalitis, inhibits the interferon (IFN) signal transduction pathway by preventing phosphorylation of Janus kinases and STAT transcription factors. Inhibition of the IFN signal cascade abrogates activation of IFN-induced genes, thus attenuating an antiviral response. We investigated the mechanism responsible for this inhibition and found that WNV infection prevents accumulation of the IFN-α receptor subunit 1 (IFNAR1). The WNV-induced depletion of IFNAR1 was conserved across multiple cell types. Our results indicated that expression of WNV nonstructural proteins resulted in activated lysosomal and proteasomal protein degradation pathways independent of the unfolded protein response (UPR). Furthermore, WNV infection did not induce serine phosphorylation, a modification on IFNAR1 that precedes its natural turnover. These data demonstrate that WNV infection results in a reduction of IFNAR1 protein through a non-canonical protein degradation pathway, and may participate in the inhibition of the IFN response.
Figures


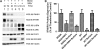
Similar articles
-
West Nile Virus NS1 Antagonizes Interferon Beta Production by Targeting RIG-I and MDA5.J Virol. 2017 Aug 24;91(18):e02396-16. doi: 10.1128/JVI.02396-16. Print 2017 Sep 15. J Virol. 2017. PMID: 28659477 Free PMC article.
-
West Nile virus differentially modulates the unfolded protein response to facilitate replication and immune evasion.J Virol. 2011 Mar;85(6):2723-32. doi: 10.1128/JVI.02050-10. Epub 2010 Dec 29. J Virol. 2011. PMID: 21191014 Free PMC article.
-
Hemagglutinin of Influenza A Virus Antagonizes Type I Interferon (IFN) Responses by Inducing Degradation of Type I IFN Receptor 1.J Virol. 2015 Dec 16;90(5):2403-17. doi: 10.1128/JVI.02749-15. J Virol. 2015. PMID: 26676772 Free PMC article.
-
Mechanisms of Immune Evasion of West Nile Virus.Rev Med Virol. 2025 May;35(3):e70042. doi: 10.1002/rmv.70042. Rev Med Virol. 2025. PMID: 40432246 Review.
-
Innate immune escape by Dengue and West Nile viruses.Curr Opin Virol. 2016 Oct;20:119-128. doi: 10.1016/j.coviro.2016.09.013. Epub 2016 Oct 25. Curr Opin Virol. 2016. PMID: 27792906 Free PMC article. Review.
Cited by
-
Finding quasi-modules of human and viral miRNAs: a case study of human cytomegalovirus (HCMV).BMC Bioinformatics. 2012 Dec 3;13:322. doi: 10.1186/1471-2105-13-322. BMC Bioinformatics. 2012. PMID: 23206407 Free PMC article.
-
The ER quality control and ER associated degradation machineries are vital for viral pathogenesis.Front Plant Sci. 2014 Mar 11;5:66. doi: 10.3389/fpls.2014.00066. eCollection 2014. Front Plant Sci. 2014. PMID: 24653727 Free PMC article. Review.
-
Innate host responses to West Nile virus: Implications for central nervous system immunopathology.World J Virol. 2013 May 12;2(2):49-56. doi: 10.5501/wjv.v2.i2.49. World J Virol. 2013. PMID: 24175229 Free PMC article. Review.
-
The Many Faces of the Flavivirus NS5 Protein in Antagonism of Type I Interferon Signaling.J Virol. 2017 Jan 18;91(3):e01970-16. doi: 10.1128/JVI.01970-16. Print 2017 Feb 1. J Virol. 2017. PMID: 27881649 Free PMC article. Review.
-
Pathogenicity and virulence of West Nile virus revisited eight decades after its first isolation.Virulence. 2021 Dec;12(1):1145-1173. doi: 10.1080/21505594.2021.1908740. Virulence. 2021. PMID: 33843445 Free PMC article.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

