Phyla- and Subtype-Selectivity of CgNa, a Na Channel Toxin from the Venom of the Giant Caribbean Sea Anemone Condylactis Gigantea
- PMID: 21833172
- PMCID: PMC3153007
- DOI: 10.3389/fphar.2010.00133
Phyla- and Subtype-Selectivity of CgNa, a Na Channel Toxin from the Venom of the Giant Caribbean Sea Anemone Condylactis Gigantea
Abstract
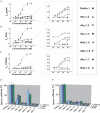
Because of their prominent role in electro-excitability, voltage-gated sodium (Na(V)) channels have become the foremost important target of animal toxins. These toxins have developed the ability to discriminate between closely related Na(V) subtypes, making them powerful tools to study Na(V) channel function and structure. CgNa is a 47-amino acid residue type I toxin isolated from the venom of the Giant Caribbean Sea Anemone Condylactis gigantea. Previous studies showed that this toxin slows the fast inactivation of tetrodotoxin-sensitive Na(V) currents in rat dorsal root ganglion neurons. To illuminate the underlying Na(V) subtype-selectivity pattern, we have assayed the effects of CgNa on a broad range of mammalian isoforms (Na(V)1.2-Na(V)1.8) expressed in Xenopus oocytes. This study demonstrates that CgNa selectively slows the fast inactivation of rNa(V)1.3/β(1), mNa(V)1.6/β(1) and, to a lesser extent, hNa(V)1.5/β(1), while the other mammalian isoforms remain unaffected. Importantly, CgNa was also examined on the insect sodium channel DmNa(V)1/tipE, revealing a clear phyla-selectivity in the efficacious actions of the toxin. CgNa strongly inhibits the inactivation of the insect Na(V) channel, resulting in a dramatic increase in peak current amplitude and complete removal of fast and steady-state inactivation. Together with the previously determined solution structure, the subtype-selective effects revealed in this study make of CgNa an interesting pharmacological probe to investigate the functional role of specific Na(V) channel subtypes. Moreover, further structural studies could provide important information on the molecular mechanism of Na(V) channel inactivation.
Keywords: inactivation; sea anemone; selectivity; sodium channel; subtype; toxin.
Figures



References
-
- Armstrong C. M. (1981). Sodium channels and gating currents. Physiol. Rev. 61, 644–683 - PubMed
-
- Black J. A., Liu S., Tanaka M., Cummins T. R., Waxman S. G. (2004). Changes in the expression of tetrodotoxin-sensitive sodium channels within dorsal root ganglia neurons in inflammatory pain. Pain 108, 237–247 - PubMed
LinkOut - more resources
Full Text Sources

