Integrin α3β1 as a breast cancer target
- PMID: 21838596
- PMCID: PMC3212412
- DOI: 10.1517/14728222.2011.609557
Integrin α3β1 as a breast cancer target
Abstract
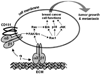
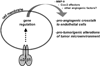
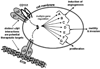
Introduction: Integrin receptors for cell adhesion to the extracellular matrix have important roles in all stages of cancer progression and metastasis. Since the integrin family was discovered in the early 1980's, many studies have identified critical adhesion and signaling functions for integrins expressed on tumor cells, endothelial cells and other cell types of the tumor microenvironment, in controlling proliferation, survival, migration and angiogenesis. In recent years, the laminin-binding integrin α3β1 has emerged as a potentially promising anti-cancer target on breast cancer cells.
Areas covered: Studies from the past decade that implicate integrins as promising anti-cancer targets and the development of integrin antagonists as anti-cancer therapeutics. Recent preclinical studies that have identified the laminin-binding integrin α3β1 as an appealing anti-cancer target and the knowledge gaps that must be closed to fully exploit this integrin as a therapeutic target for breast cancer.
Expert opinion: Although the tumor-promoting functions of α3β1 implicate this integrin as a promising therapeutic target on breast cancer cells, successful exploitation of this integrin as an anti-cancer target will require a better understanding of the molecular mechanisms whereby it regulates specific tumor cell behaviors and the identification of the most appropriate α3β1 functions to antagonize on breast cancer cells.
Figures
References
-
- Weiner LM. Building better magic bullets-improving unconjugated monoclonal antibody therapy for cancer. Nat Rev Cancer. 2007;7:701–706. - PubMed
-
- Hurwitz H, Fehrenbacher L, Novotny W, et al. Bevacizumab plus irinotecan, fluorouracil, and leucovorin for metastatic colorectal cancer. N Engl J Med. 2004;350:2335–2342. - PubMed
-
- Slamon DJ, Leyland-Jones B, Shak S, et al. Use of chemotherapy plus a monoclonal antibody against HER2 for metastatic breast cancer that overexpresses HER2. N Engl J Med. 2001;344:783–792. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous
