Virion glycosylation governs integrity and infectivity of infectious pancreatic necrosis virus
- PMID: 21838710
- PMCID: PMC7197461
- DOI: 10.1111/j.1365-2761.2011.01280.x
Virion glycosylation governs integrity and infectivity of infectious pancreatic necrosis virus
Abstract
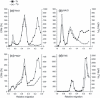
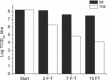
The possible importance of the O-linked glycosylation in virion stability and infectivity of infectious pancreatic necrosis virus (IPNV) was analysed. Enzymatic treatment with O-glycosidase of radiolabelled virions under different ionic conditions, to allow for possible alternative exposure of glycosidic enzyme cleavage sites, did not alter the specific infectivity of virions re-isolated after rate-zonal centrifugation in glycerol gradients. As an alternative method to assess the significance of carbohydrates in IPNV integrity, periodate oxidation in the presence of an aldehyde quencher was chosen. Following re-isolation of viruses, a 3-5 (10)log-unit reduction in specific infectivity was revealed and, at higher concentrations, a total disruption or virion aggregation was observed. The loss of infectivity of intact virions was not because of a lack of attachment to cells. Additionally, re-evaluation of reading values from UV-spectra of purified IPNV yielded a specific infectivity of 3 × 10(11) TCID(50)-units mg(-1) of protein and a ratio of 40 virions per TCID(50)-unit in the CHSE-214 cell system.
© 2011 Blackwell Publishing Ltd.
Figures

Similar articles
-
Infectious pancreatic necrosis virus: identification of a VP3-containing ribonucleoprotein core structure and evidence for O-linked glycosylation of the capsid protein VP2.J Virol. 1999 Apr;73(4):3484-90. doi: 10.1128/JVI.73.4.3484-3490.1999. J Virol. 1999. PMID: 10074207 Free PMC article.
-
Genome assembly and particle maturation of the birnavirus infectious pancreatic necrosis virus.J Virol. 2004 Dec;78(24):13829-38. doi: 10.1128/JVI.78.24.13829-13838.2004. J Virol. 2004. PMID: 15564491 Free PMC article.
-
In infectious pancreatic necrosis virus carrier Atlantic salmon, Salmo salar L., post-smolts, almost all kidney macrophages ex vivo contain a low level of non-replicating virus.J Fish Dis. 2006 Jan;29(1):43-8. doi: 10.1111/j.1365-2761.2005.00680.x. J Fish Dis. 2006. PMID: 16351697
-
Autophagy induced by infectious pancreatic necrosis virus promotes its multiplication in the Chinook salmon embryo cell line CHSE-214.Fish Shellfish Immunol. 2020 Feb;97:375-381. doi: 10.1016/j.fsi.2019.12.067. Epub 2019 Dec 23. Fish Shellfish Immunol. 2020. PMID: 31874298
-
Molecular determinants of the ratio of inert to infectious virus particles.Prog Mol Biol Transl Sci. 2015;129:285-326. doi: 10.1016/bs.pmbts.2014.10.012. Epub 2014 Dec 1. Prog Mol Biol Transl Sci. 2015. PMID: 25595808 Free PMC article. Review.
Cited by
-
The Infectious Pancreatic Necrosis Virus (IPNV) and its Virulence Determinants: What is Known and What Should be Known.Pathogens. 2020 Feb 4;9(2):94. doi: 10.3390/pathogens9020094. Pathogens. 2020. PMID: 32033004 Free PMC article. Review.
References
-
- Ahuja D., Saenz‐Robles M.T. & Pipas J.M. (2005) SV40 large T antigen targets multiple cellular pathways to elicit cellular transformation. Oncogene 24, 7729–7745. - PubMed
-
- Banerjee M. & Johnson J.E. (2008) Activation, exposure and penetration of virally encoded, membrane‐active polypeptides during non‐enveloped virus entry. Current Protein and Peptide Science 9, 116–127. - PubMed
-
- Brown F. (1986) The classification and nomenclature of viruses: summary of results of meetings of the International Committee on Taxonomy of Viruses in Sendai, September 1984. Intervirology 25, 141–143. - PubMed
-
- Burrage T., Kramer E. & Brown F. (2000) Structural differences between foot‐and‐mouth disease and poliomyelitis viruses influence their inactivation by aziridines. Vaccine 18, 2454–2461. - PubMed
-
- Chang N., Macdonald R.D. & Yamamoto T. (1978) Purification of infectious pancreatic necrosis (IPN) virus and comparison of polypeptide composition of different isolates. Canadian Journal of Microbiology 24, 19–27. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials

