Government use licenses in Thailand: an assessment of the health and economic impacts
- PMID: 21838926
- PMCID: PMC3176479
- DOI: 10.1186/1744-8603-7-28
Government use licenses in Thailand: an assessment of the health and economic impacts
Abstract
Background: Between 2006 and 2008, Thailand's Ministry of Public Health (MOPH) granted government use licenses for seven patented drugs in order to improve access to these essential treatments. The decision to grant the government use licenses was contentious both within and beyond the country. In particular, concerns were highlighted that the negative consequences might outweigh the expected benefits of the policy. This study conducted assessments of the health and economic implications of these government use licenses.
Methods: The health and health-related economic impacts were quantified in terms of i) Quality Adjusted Life Years (QALYs) gained and ii) increased productivity in US dollars (USD) as a result of the increased access to drugs. The study adopted a five-year timeframe for the assessment, commencing from the time of the grant of the government use licenses. Empirical evidence gathered from national databases was used to assess the changes in volume of exports after US Generalized System of Preferences (GSP) withdrawal and level of foreign direct investment (FDI).
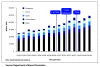
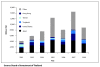
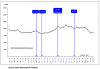
Results: As a result of the granting of the government use licenses, an additional 84,158 patients were estimated to have received access to the seven drugs over five years. Health gains from the use of the seven drugs compared to their best alternative accounted for 12,493 QALYs gained, which translates into quantifiable incremental benefits to society of USD132.4 million. The government use license on efavirenze was found to have the greatest benefit. In respect of the country's economy, the study found that Thailand's overall exports increased overtime, although exports of the three US GSP withdrawal products to the US did decline. There was also found to be no relationship between the government use licenses and the level of foreign investment over the period 2002 to 2008.
Conclusions: The public health benefits of the government use licenses were generally positive. Specifically, the policy helped to increase access to patented drugs, while the impact of the US GSP withdrawal did not adversely affect the overall export status. Because the levels of benefit gained from the government use licenses varied widely between the seven drugs, depending on several factors, this study makes recommendations for the future implementation of the policy in order to maximise benefits.
Figures

References
-
- World Trade Organization. Agreement on trade-related aspects of intellectual property rights. Geneva: World Trade Organization; 2001. http://www.wto.org/english/docs_e/legal_e/27-trips.pdf - PubMed
-
- Correa CM. Bilateralism in intellectual property: defeating the WTO system for access to medicines. Case Western Reserve Journal of International Law. 2004;36:79–94.
-
- 't Hoen E. TRIPS, pharmaceutical patents, and access to essential medicines: a long way from Seattle to Doha. Chicago Journal of International Law. 2002;3:27–46. - PubMed
-
- World Health Organization. International Trade and Health: a reference guide. New Delhi, India: World Health Organization, Regional Office for South-East Asia; 2009.
LinkOut - more resources
Full Text Sources
Research Materials

