Desiccating stress induces CD4+ T-cell-mediated Sjögren's syndrome-like corneal epithelial apoptosis via activation of the extrinsic apoptotic pathway by interferon-γ
- PMID: 21843497
- PMCID: PMC3181354
- DOI: 10.1016/j.ajpath.2011.06.030
Desiccating stress induces CD4+ T-cell-mediated Sjögren's syndrome-like corneal epithelial apoptosis via activation of the extrinsic apoptotic pathway by interferon-γ
Abstract
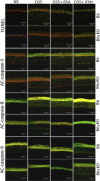
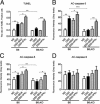
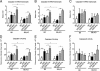
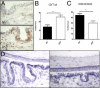
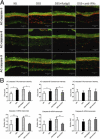
We investigated the role of CD4(+) T-cell-produced interferon (IFN)-γ on corneal epithelial apoptosis in a murine desiccating stress (DS) model that resembles Sjögren's syndrome. The DS model was generated in C57BL/6 (B6) and B6 IFN-γ-knockout (B6γKO) mice. Adoptive transfer of CD4(+) T cells from DS-exposed donor to recombination activating gene (RAG)-1(-/-) recipient mice and topical neutralization of IFN-γ were performed to determine whether IFN-γ produced by pathogenic CD4(+) T cells promotes corneal epithelial apoptosis. Apoptosis in corneal epithelia was assessed by evaluating the expression and activity of caspases 3, 8, and 9. The activation of caspase-8 mediated increased corneal epithelial apoptosis in B6 mice after DS, and this was exacerbated by subconjunctival IFN-γ injection. B6γKO mice were resistant to DS-induced apoptosis; however, B6γKO mice receiving IFN-γ developed apoptosis similar to that observed in B6 wild-type mice. Adoptive transfer of CD4(+) T cells from donors subjected to DS increased corneal epithelial apoptosis via activation of caspase-8 in recipients, similar to that in the donor mice. Topical neutralization of IFN-γ in adoptive transfer recipients decreased corneal epithelial apoptosis. DS, IFN-γ administration, or CD4(+) T-cell adoptive transfer had no effect on the expression and activation of the intrinsic apoptosis mediator, caspase-9. CD4(+) T-cell-produced IFN-γ plays a pivotal role in DS-induced corneal epithelial apoptosis via activation of the extrinsic apoptotic pathway.
Copyright © 2011 American Society for Investigative Pathology. Published by Elsevier Inc. All rights reserved.
Figures
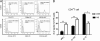
References
-
- Pflugfelder S.C., Tseng S.C.G., Sanabria O., Kell H., Garcia C.G., Felix C., Feuer W., Reis B.L. Evaluation of subjective assessments and objective diagnostic tests for diagnosing tear-film disorders known to cause ocular irritation. Cornea. 1998;17:38–56. - PubMed
-
- de Paiva C.S., Hwang C.S., Pitcher J.D., III, Pangelinan S.B., Rahimy E., Chen W., Yoon K.C., Farley W.J., Niederkorn J.Y., Stern M.E., Li D.Q., Pflugfelder S.C. Age-related T-cell cytokine profile parallels corneal disease severity in Sjogren's syndrome-like keratoconjunctivitis sicca in CD25KO mice. Rheumatology (Oxford) 2010;49:246–258. - PMC - PubMed
-
- De Paiva C.S., Villarreal A.L., Corrales R.M., Rahman H.T., Chang V.Y., Farley W.J., Stern M.E., Niederkorn J.Y., Li D.Q., Pflugfelder S.C. Dry eye-induced conjunctival epithelial squamous metaplasia is modulated by interferon-gamma. Invest Ophthalmol Vis Sci. 2007;48:2553–2560. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Research Materials

