Withdrawing PPI therapy after healing esophagitis does not worsen symptoms or cause persistent hypergastrinemia: analysis of dexlansoprazole MR clinical trial data
- PMID: 21844923
- PMCID: PMC3209587
- DOI: 10.1038/ajg.2011.220
Withdrawing PPI therapy after healing esophagitis does not worsen symptoms or cause persistent hypergastrinemia: analysis of dexlansoprazole MR clinical trial data
Abstract
Objectives: Withdrawal of proton pump inhibitors (PPIs) may induce symptoms in healthy volunteers, suggesting that discontinuing PPI therapy induces acid-peptic disease. Similar assessments in patients with documented acid-related disorders are lacking.
Methods: We performed a retrospective analysis of data from 287 Helicobacter pylori-negative erosive esophagitis (EE) patients healed after 4 or 8 weeks of therapy with dexlansoprazole modified release (MR) or lansoprazole, and then randomized to placebo in 6-month maintenance trials. We compared serum gastrin levels and 24-h heartburn severity before enrollment in the healing trials (baseline) and after receiving placebo in the 6-month maintenance trials.
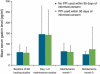
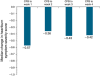
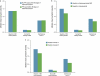
Results: Mean gastrin values at maintenance months 1 and 3 were essentially unchanged (median changes, 1.0 and -1.0 pg/ml), showing that gastrin normalized within 1 month of discontinuing PPIs and remained flat. Mean heartburn severity at maintenance month 1 was <1 on a 5-point scale (1=mild) and significantly lower than at baseline (median decrease, 0.41 points; P≤0.001). Heartburn severity in patients healed at week 4 or 8 with either PPI was generally similar, suggesting that neither longer exposure nor more potent therapy was associated with rebound. In those with month 2 data, mean heartburn severity at months 1 and 2 was significantly lower than baseline (median decrease, 0.54 and 0.58 points; both P<0.001), indicating an ongoing symptom response for 2 months after PPI withdrawal.
Conclusions: In H. pylori-negative EE patients, there was no indication of recurring heartburn symptom worsening beyond baseline levels within 2 months of discontinuing 4-8 weeks of PPI therapy.
Figures
Comment in
-
Withdrawing PPI therapy: response to Metz et al.Am J Gastroenterol. 2012 Feb;107(2):325-6; author reply 326. doi: 10.1038/ajg.2011.391. Am J Gastroenterol. 2012. PMID: 22306953 No abstract available.
Similar articles
-
The effects of increasing body mass index on heartburn severity, frequency and response to treatment with dexlansoprazole or lansoprazole.Aliment Pharmacol Ther. 2013 Apr;37(8):810-8. doi: 10.1111/apt.12270. Epub 2013 Mar 4. Aliment Pharmacol Ther. 2013. PMID: 23451835 Free PMC article.
-
Distinguishing the impact of dexlansoprazole on heartburn vs. regurgitation in patients with gastro-oesophageal reflux disease.Aliment Pharmacol Ther. 2013 Nov;38(10):1303-11. doi: 10.1111/apt.12504. Epub 2013 Sep 30. Aliment Pharmacol Ther. 2013. PMID: 24118079 Clinical Trial.
-
Clinical trial: dexlansoprazole MR, a proton pump inhibitor with dual delayed-release technology, effectively controls symptoms and prevents relapse in patients with healed erosive oesophagitis.Aliment Pharmacol Ther. 2009 Apr 1;29(7):742-54. doi: 10.1111/j.1365-2036.2009.03954.x. Epub 2009 Feb 7. Aliment Pharmacol Ther. 2009. PMID: 19210298 Clinical Trial.
-
Dexlansoprazole in the treatment of esophagitis and gastroesophageal reflux disease.Ann Pharmacother. 2010 May;44(5):871-7. doi: 10.1345/aph.1M685. Epub 2010 Apr 6. Ann Pharmacother. 2010. PMID: 20371754 Review.
-
Dexlansoprazole modified release: in erosive oesophagitis and non-erosive reflux disease.Drugs. 2010 Aug 20;70(12):1593-601. doi: 10.2165/11295960-000000000-00000. Drugs. 2010. PMID: 20687622 Review.
Cited by
-
Consumption of medicines used for gastric acid-related disorders in Australia and South Korea: a cross-country comparison.Eur J Clin Pharmacol. 2020 Apr;76(4):547-555. doi: 10.1007/s00228-019-02798-8. Epub 2019 Dec 10. Eur J Clin Pharmacol. 2020. PMID: 31822956
-
Proton pump inhibitors for the treatment of patients with erosive esophagitis and gastroesophageal reflux disease: current evidence and safety of dexlansoprazole.Clin Exp Gastroenterol. 2016 Jul 13;9:163-72. doi: 10.2147/CEG.S91602. eCollection 2016. Clin Exp Gastroenterol. 2016. PMID: 27471402 Free PMC article. Review.
-
Deprescribing proton pump inhibitors: Evidence-based clinical practice guideline.Can Fam Physician. 2017 May;63(5):354-364. Can Fam Physician. 2017. PMID: 28500192 Free PMC article.
-
Randomised trial of the effect of a gastrin/CCK2 receptor antagonist on esomeprazole-induced hypergastrinaemia: evidence against rebound hyperacidity.Eur J Clin Pharmacol. 2017 Feb;73(2):129-139. doi: 10.1007/s00228-016-2150-x. Epub 2016 Oct 29. Eur J Clin Pharmacol. 2017. PMID: 27796466 Clinical Trial.
-
Prescribing proton pump inhibitors: is it time to pause and rethink?Drugs. 2012 Mar 5;72(4):437-45. doi: 10.2165/11599320-000000000-00000. Drugs. 2012. PMID: 22356286 Review.
References
-
- Kahrilas PJ, Shaheen NJ, Vaezi MF, et al. American Gastroenterological Association Medical Position Statement on the management of gastroesophageal reflux disease Gastroenterology 20081351383–1391.1391 e1–5. - PubMed
-
- Donnellan C, Sharma N, Preston C, et al. Medical treatments for the maintenance therapy of reflux oesophagitis and endoscopic negative reflux disease. Cochrane Database Syst Rev. 2004. p. CD003245. - PubMed
-
- Sandvik AK, Brenna E, Waldum HL. Review article: the pharmacological inhibition of gastric acid secretion--tolerance and rebound. Aliment Pharmacol Ther. 1997;11:1013–1018. - PubMed
-
- Waldum HL, Qvigstad G, Fossmark R, et al. Rebound acid hypersecretion from a physiological, pathophysiological and clinical viewpoint. Scand J Gastroenterol. 2010;45:389–394. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

