Predictive value of interferon-γ release assays for incident active tuberculosis: a systematic review and meta-analysis
- PMID: 21846592
- PMCID: PMC3568693
- DOI: 10.1016/S1473-3099(11)70210-9
Predictive value of interferon-γ release assays for incident active tuberculosis: a systematic review and meta-analysis
Abstract
Background: We aimed to assess whether interferon-γ release assays (IGRAs) can predict the development of active tuberculosis and whether the predictive ability of these tests is better than that of the tuberculin skin test (TST).
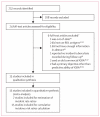
Methods: Longitudinal studies of the predictive value for active tuberculosis of in-house or commercial IGRAs were identified through searches of PubMed, Embase, Biosis, and Web of Science and complementary manual searches up to June 30, 2011. Eligible studies included adults or children, with or without HIV, who were free of active tuberculosis at study baseline. We summarised incidence rates in forest plots and pooled data with random-effects models when appropriate. We calculated incidence rate ratios (IRR) for rates of disease progression in IGRA-positive versus IGRA-negative individuals.
Findings: 15 studies had a combined sample size of 26 680 participants. Incidence of tuberculosis during a median follow-up of 4 years (IQR 2-6), even in IGRA-positive individuals, was 4-48 cases per 1000 person-years. Seven studies with no possibility of incorporation bias and reporting baseline stratification on the basis of IGRA results showed a moderate association between positive results and subsequent tuberculosis (pooled unadjusted IRR 2·10, 95% CI 1·42-3·08). Compared with test-negative results, IGRA-positive and TST-positive results were much the same with regard to the risk of tuberculosis (pooled IRR in the five studies that used both was 2·11 [95% CI 1·29-3·46] for IGRA vs 1·60 [0·94-2·72] for TST at the 10 mm cutoff). However, the proportion of IGRA-positive individuals in seven of 11 studies that assessed both IGRAs and TST was generally lower than TST-positive individuals.
Interpretation: Neither IGRAs nor the TST have high accuracy for the prediction of active tuberculosis, although use of IGRAs in some populations might reduce the number of people considered for preventive treatment. Until more predictive biomarkers are identified, existing tests for latent tuberculosis infection should be chosen on the basis of relative specificity in different populations, logistics, cost, and patients' preferences rather than on predictive ability alone.
Funding: Special Programme for Research and Training in Tropical Diseases (WHO), Wellcome Trust, Canadian Institutes of Health Research, UK Medical Research Council, and the European and Developing Countries Clinical Trials Partnership.
Copyright © 2012 Elsevier Ltd. All rights reserved.
Figures




Comment in
-
Tests for prediction of active tuberculosis.Lancet Infect Dis. 2012 Jan;12(1):6-8. doi: 10.1016/S1473-3099(11)70215-8. Epub 2011 Aug 16. Lancet Infect Dis. 2012. PMID: 21846593 No abstract available.
-
Interferon-γ release assays for prediction of tuberculosis.Lancet Infect Dis. 2012 Aug;12(8):584. doi: 10.1016/S1473-3099(12)70169-X. Lancet Infect Dis. 2012. PMID: 22835892 No abstract available.
-
Tests for latent tuberculosis infection and isoniazid preventive therapy.Lancet Infect Dis. 2012 Nov;12(11):827-8. doi: 10.1016/S1473-3099(12)70221-9. Lancet Infect Dis. 2012. PMID: 23099076 No abstract available.
References
-
- Dye C, Scheele S, Dolin P, Pathania V, Raviglione MC. Consensus statement: global burden of tuberculosis—estimated incidence, prevalence, and mortality by country (WHO global surveillance and monitoring project) JAMA. 1999;282:677–86. - PubMed
-
- Woldehanna S, Volmink J. Treatment of latent tuberculosis infection in HIV infected persons. Cochrane Database Syst Rev. 2004;1:CD000171. - PubMed
-
- Diel R, Loddenkemper R, Nienhaus A. Evidence-based comparison of commercial interferon-gamma release assays for detecting active tuberculosis: a metaanalysis. Chest. 2009;137:952–68. - PubMed
-
- Pai M, Riley LW, Colford JM., Jr. Interferon-gamma assays in the immunodiagnosis of tuberculosis: a systematic review. Lancet Infect Dis. 2004;4:761–76. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources

