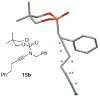
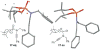Introducing a new class of N-phosphoryl ynamides via Cu(I)-catalyzed amidations of alkynyl bromides
- PMID: 21848304
- PMCID: PMC3170467
- DOI: 10.1021/ol201947b
Introducing a new class of N-phosphoryl ynamides via Cu(I)-catalyzed amidations of alkynyl bromides
Abstract
We describe here the first synthesis of N-phosphoryl ynamides featuring C- and P-chirality via copper(I)-catalyzed amidative cross-couplings between phosphoramidates and phosphordiamidates with alkynyl bromides. Also featured is a tandem aza-Claisen-hetero-[2+2] cycloaddition for the synthesis of N-phosphoryl azetidin-2-imines.
© 2011 American Chemical Society
Figures
Similar articles
-
Synthesis of cyclopentenimines from N-allyl ynamides via a tandem aza-Claisen rearrangement-carbocyclization sequence.J Org Chem. 2013 Jun 21;78(12):6233-44. doi: 10.1021/jo400960e. Epub 2013 Jun 10. J Org Chem. 2013. PMID: 23718841 Free PMC article.
-
Copper(II)-catalyzed amidations of alkynyl bromides as a general synthesis of ynamides and Z-enamides. An intramolecular amidation for the synthesis of macrocyclic ynamides.J Org Chem. 2006 May 26;71(11):4170-7. doi: 10.1021/jo060230h. J Org Chem. 2006. PMID: 16709057
-
Palladium-catalyzed carbonylation reactions of aryl bromides at atmospheric pressure: a general system based on Xantphos.J Org Chem. 2008 Sep 19;73(18):7102-7. doi: 10.1021/jo801279r. Epub 2008 Aug 23. J Org Chem. 2008. PMID: 18720970 Free PMC article.
-
Copper catalyzed decarboxylative alkynylation of quaternary α-cyano acetate salts.Org Lett. 2013 Apr 5;15(7):1472-5. doi: 10.1021/ol400197y. Epub 2013 Mar 8. Org Lett. 2013. PMID: 23472715
-
Cu-catalyzed azide-alkyne cycloaddition.Chem Rev. 2008 Aug;108(8):2952-3015. doi: 10.1021/cr0783479. Chem Rev. 2008. PMID: 18698735 Review. No abstract available.
Cited by
-
Synthesis of cyclopentenimines from N-allyl ynamides via a tandem aza-Claisen rearrangement-carbocyclization sequence.J Org Chem. 2013 Jun 21;78(12):6233-44. doi: 10.1021/jo400960e. Epub 2013 Jun 10. J Org Chem. 2013. PMID: 23718841 Free PMC article.
-
Asymmetric synthesis with ynamides: unique reaction control, chemical diversity and applications.Chem Soc Rev. 2020 Dec 7;49(23):8543-8583. doi: 10.1039/d0cs00769b. Epub 2020 Oct 19. Chem Soc Rev. 2020. PMID: 33073285 Free PMC article.
-
Batch and flow photochemical benzannulations based on the reaction of ynamides and diazo ketones. Application to the synthesis of polycyclic aromatic and heteroaromatic compounds.J Org Chem. 2013 Nov 15;78(22):11450-69. doi: 10.1021/jo402010b. Epub 2013 Oct 28. J Org Chem. 2013. PMID: 24116731 Free PMC article.
-
Carbocyclization cascades of allyl ketenimines via aza-Claisen rearrangements of N-phosphoryl-N-allyl-ynamides.Org Lett. 2012 Apr 6;14(7):1768-71. doi: 10.1021/ol300366e. Epub 2012 Mar 13. Org Lett. 2012. PMID: 22414252 Free PMC article.
-
An intramolecular [2 + 2] cycloaddition of ketenimines via palladium-catalyzed rearrangements of N-allyl-ynamides.Org Lett. 2012 Jun 15;14(12):3214-7. doi: 10.1021/ol3013233. Epub 2012 Jun 5. Org Lett. 2012. PMID: 22667819 Free PMC article.
References
-
-
For current leading reviews on ynamides, see Evano G, Coste A, Jouvin K. Angew Chem Int Ed. 2010;49:2840. DeKorver KA, Li H, Lohse AG, Hayashi R, Lu Z, Zhang Y, Hsung RP. Chem Rev. 2010;110:5064.
-
-
-
For ynamide papers published in 2011 alone, see: Balieu S, Toutah K, Carro L, Chamoreau L-M, Rouselière H, Courillon C. Tetrahedron Lett. 2011;52:2876. Fadel A, Legrand F, Evano G, Rabasso N. Adv Synth Catal. 2011;353:263. Schotes C, Mezzetti A. Angew Chem. 2011;123:3128. Barbazanges M, Meyer C, Cossy J, Turner P. Chem–Eur J. 2011;17:4480. Pizzetti M, Russo A, Petricci E. Chem–Eur J. 2011;17:4523. Garcia P, Evanno Y, George P, Sevrin M, Ricci G, Malacria M, Aubert C, Gandon V. Org Lett. 2011;13:2030. Kramer S, Friis SD, Xin Z, Odabachian Y, Skrydstrup T. Org Lett. 2011;13:1750. Li C, Zhang L. Org Lett. 2011;13:1738. Wang YP, Danheiser RL. Tetrahedron Lett. 2011;52:2111. Mak XY, Crombie AL, Danheiser RL. J Org Chem. 2011;76:1852. Davies PW, Cremonesi A, Martin N. Chem Commun. 2011:379. Xu C-F, Mei Xu M, Jia YX, Li C-Y. Org Lett. 2011;13:1556. Chen Z, Zheng D, Wu J. Org Lett. 2011;13:848. Shindoh N, Takemoto Y, Takasu K. Heterocycles. 2011;82:1133. Saito N, Katayama T, Saito K. Heterocycles. 2011;82:1181. Kramer S, Odabachian Y, Overgaard J, Rottländer M, Gagosz F, Skrydstrup T. Angew Chemie. 2011;123:5196. Nissen F, Richard V, Alayrac C, Witulski B. Chem Commun. 2011;47:6656. Hashmi ASK, Schuster AM, Zimmer M, Rominger F. Chem–Eur J. 2011;17:5511. Gilboa N, Wang H, Houk KN, Marek I. Chem–Eur J. 2011;17:8000. Chemla F, Dulong F, Ferreira F, Nüllen MP, Pérez-Luna A. Synthesis. 2011;9:1347. Saito N, Saito K, Shiro M, Sato Y. Org Lett. 2011;13:2718.
-
-
-
For leading reviews on the synthesis of ynamides, see: Tracey MR, Hsung RP, Antoline JA, Kurtz KCM, Shen L, Slafer BW, Zhang Y. In: Science of Synthesis, Houben-Weyl Methods of Molecular Transformations. Weinreb Steve M., editor. Chapter 21.4. Georg Thieme Verlag KG; Stuttgart, Germany: 2005. Mulder JA, Kurtz KCM, Hsung RP. Synlett. 2003:1379.
-
-
- Fromont C, Masson S. Tetrahedron. 1999;55:5405.
-
- Sueda T, Oshima A, Teno N. Org Lett. 2011;13:3996. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources