Evaluation of In-Labeled Cyclic RGD Peptides: Effects of Peptide and Linker Multiplicity on Their Tumor Uptake, Excretion Kinetics and Metabolic Stability
- PMID: 21850213
- PMCID: PMC3157017
- DOI: 10.7150/thno/v01p0322
Evaluation of In-Labeled Cyclic RGD Peptides: Effects of Peptide and Linker Multiplicity on Their Tumor Uptake, Excretion Kinetics and Metabolic Stability
Abstract
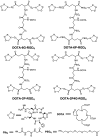
Purpose: The purpose of this study was to demonstrate the valence of cyclic RGD peptides, P-RGD (PEG(4)-c(RGDfK): PEG(4) = 15-amino-4,710,13-tetraoxapentadecanoic acid), P-RGD(2) (PEG(4)-E[c(RGDfK)](2), 2P-RGD(4) (E{PEG(4)-E[c(RGDfK)](2)}(2), 2P4G-RGD(4) (E{PEG(4)-E[G(3)-c(RGDfK)](2)}(2): G(3) = Gly-Gly-Gly) and 6P-RGD(4) (E{PEG(4)-E[PEG(4)-c(RGDfK)](2)}(2)) in binding to integrin α(v)β(3), and to assess the impact of peptide and linker multiplicity on biodistribution properties, excretion kinetics and metabolic stability of their corresponding (111)In radiotracers.
Methods: Five new RGD peptide conjugates (DOTA-P-RGD (DOTA =1,4,7,10-tetraazacyclododecane-1,4,7,10-tetracetic acid), DOTA-P-RGD(2), DOTA-2P-RGD(4), DOTA-2P4G-RGD(4), DOTA-6P-RGD(4)), and their (111)In complexes were prepared. The integrin α(v)β(3) binding affinity of cyclic RGD conjugates were determined by a competitive displacement assay against (125)I-c(RGDyK) bound to U87MG human glioma cells. Biodistribution, planar imaging and metabolism studies were performed in athymic nude mice bearing U87MG human glioma xenografts.
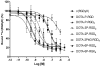
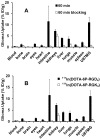
Results: The integrin α(v)β(3) binding affinity of RGD conjugates follows the order of: DOTA-6P-RGD(4) (IC(50) = 0.3 ± 0.1 nM) ~ DOTA-2P4G-RGD(4) (IC(50) = 0.2 ± 0.1 nM) ~ DOTA-2P-RGD(4) (IC(50) = 0.5 ± 0.1 nM) > DOTA-3P-RGD(2) (DOTA-PEG(4)-E[PEG(4)-c(RGDfK)](2): IC(50) = 1.5 ± 0.2 nM) > DOTA-P-RGD(2) (IC(50) = 5.0 ± 1.0 nM) >> DOTA-P-RGD (IC(50) = 44.3 ± 3.5 nM) ~ c(RGDfK) (IC(50) = 49.9 ± 5.5 nM) >> DOTA-6P-RGK(4) (IC(50) = 437 ± 35 nM). The fact that DOTA-6P-RGK(4) had much lower integrin α(v)β(3) binding affinity than DOTA-6P-RGD(4) suggests that the binding of DOTA-6P-RGD(4) to integrin α(v)β(3) is RGD-specific. This conclusion is consistent with the lower tumor uptake for (111)In(DOTA-6P-RGK(4)) than that for (111)In(DOTA-6P-RGD(4)). It was also found that the G(3) and PEG(4) linkers between RGD motifs have a significant impact on the integrin α(v)β(3)-targeting capability, biodistribution characteristics, excretion kinetics and metabolic stability of (111)In-labeled cyclic RGD peptides.
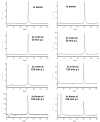
Conclusion: On the basis of their integrin α(v)β(3) binding affinity and tumor uptake of their corresponding (111)In radiotracers, it was conclude that 2P-RGD(4), 2P4G-RGD(4) and 6P-RGD(4) are most likely bivalent in binding to integrin α(v)β(3), and extra RGD motifs might contribute to the long tumor retention times of (111)In(DOTA-2P-RGD(4)),( 111)In(DOTA-2P4G-RGD(4)) and (111)In(DOTA-6P-RGD(4)) than that of (111)In(DOTA-3P-RGD(3)) at 72 h p.i. Among the (111)In-labeled cyclic RGD tetramers evaluated in the glioma model, (111)In(DOTA-2P4G-RGD(4)) has very high tumor uptake with the best tumor/kidney and tumor/liver ratios, suggesting that (90)Y(DOTA-2P4G-RGD(4)) and (177)Lu(DOTA-2P4G-RGD(4)) might have the potential for targeted radiotherapy of integrin α(v)β(3)-positive tumors.
Keywords: 111In-labeled cyclic RGD peptides; integrin αvβ3; tumor imaging.
Conflict of interest statement
Conflict of Interest: The authors have declared that no conflict of interest exists.
Figures



Similar articles
-
111In-Labeled 1,4,7,10-tetraazacyclododecane-1,4,7,10-tetracetic acid-Glu{PEG4-Glu[cyclo(Lys(Gly-Gly-Gly)-Arg-Gly-Asp-d-Phe)]-cyclo(Lys(Gly-Gly-Gly)-Arg-Gly-Asp-d-Phe)}-{PEG4-Glu[cyclo(Lys(Gly-Gly-Gly)-Arg-Gly-Asp-d-Phe)]-cyclo(Lys(Gly-Gly-Gly)-Arg-Gly-Asp-d-Phe)} (PEG4 = 15 amino-4,7,10,13-tetraoxapentadecanoic acid).2012 Feb 23 [updated 2012 Mar 22]. In: Molecular Imaging and Contrast Agent Database (MICAD) [Internet]. Bethesda (MD): National Center for Biotechnology Information (US); 2004–2013. 2012 Feb 23 [updated 2012 Mar 22]. In: Molecular Imaging and Contrast Agent Database (MICAD) [Internet]. Bethesda (MD): National Center for Biotechnology Information (US); 2004–2013. PMID: 22457888 Free Books & Documents. Review.
-
111In-Labeled 1,4,7,10-tetraazacyclododecane-1,4,7,10-tetracetic acid-Glu{PEG4-Glu[cyclo(Lys(PEG4)-Arg-Gly-Asp-d-Phe)]-cyclo(Lys(PEG4)-Arg-Gly-Asp-d-Phe)}-{PEG4-Glu[cyclo(Lys(PEG4)-Arg-Gly-Asp-d-Phe)]-cyclo(Lys(PEG4)-Arg-Gly-Asp-d-Phe)} (PEG4 = 15 amino-4,7,10,13-tetraoxapentadecanoic acid).2012 Feb 23 [updated 2012 Mar 22]. In: Molecular Imaging and Contrast Agent Database (MICAD) [Internet]. Bethesda (MD): National Center for Biotechnology Information (US); 2004–2013. 2012 Feb 23 [updated 2012 Mar 22]. In: Molecular Imaging and Contrast Agent Database (MICAD) [Internet]. Bethesda (MD): National Center for Biotechnology Information (US); 2004–2013. PMID: 22457890 Free Books & Documents. Review.
-
111In-Labeled 1,4,7,10-tetraazacyclododecane-1,4,7,10-tetracetic acid-Glu{PEG4-Glu[cyclo(Lys-Arg-Gly-Asp-d-Phe)]-cyclo(Lys-Arg-Gly-Asp-d-Phe)}-{PEG4-Glu[cyclo(Lys-Arg-Gly-Asp-d-Phe)]-cyclo(Lys-Arg-Gly-Asp-d-Phe)} (PEG4 = 15 amino-4,710,13-tetraoxapentadecanoic acid).2012 Feb 23 [updated 2012 Mar 22]. In: Molecular Imaging and Contrast Agent Database (MICAD) [Internet]. Bethesda (MD): National Center for Biotechnology Information (US); 2004–2013. 2012 Feb 23 [updated 2012 Mar 22]. In: Molecular Imaging and Contrast Agent Database (MICAD) [Internet]. Bethesda (MD): National Center for Biotechnology Information (US); 2004–2013. PMID: 22457887 Free Books & Documents. Review.
-
Improving tumor uptake and pharmacokinetics of (64)Cu-labeled cyclic RGD peptide dimers with Gly(3) and PEG(4) linkers.Bioconjug Chem. 2009 Apr;20(4):750-9. doi: 10.1021/bc800455p. Bioconjug Chem. 2009. PMID: 19320477 Free PMC article.
-
Improving tumor-targeting capability and pharmacokinetics of (99m)Tc-labeled cyclic RGD dimers with PEG(4) linkers.Mol Pharm. 2009 Jan-Feb;6(1):231-45. doi: 10.1021/mp800150r. Mol Pharm. 2009. PMID: 19067525 Free PMC article.
Cited by
-
Evaluation of K(HYNIC)(2) as a bifunctional chelator for (99m)Tc-labeling of small biomolecules.Bioconjug Chem. 2013 Apr 17;24(4):701-11. doi: 10.1021/bc3006896. Epub 2013 Apr 8. Bioconjug Chem. 2013. PMID: 23521129 Free PMC article.
-
Palmitic Acid-Conjugated Radiopharmaceutical for Integrin αvβ3-Targeted Radionuclide Therapy.Pharmaceutics. 2022 Jun 23;14(7):1327. doi: 10.3390/pharmaceutics14071327. Pharmaceutics. 2022. PMID: 35890224 Free PMC article.
-
Vascular Targeting of a Gold Nanoparticle to Breast Cancer Metastasis.J Pharm Sci. 2015 Aug;104(8):2600-10. doi: 10.1002/jps.24518. Epub 2015 Jun 2. J Pharm Sci. 2015. PMID: 26036431 Free PMC article.
-
Monitoring tumor response to linifanib therapy with SPECT/CT using the integrin αvβ3-targeted radiotracer 99mTc-3P-RGD2.J Pharmacol Exp Ther. 2013 Aug;346(2):251-8. doi: 10.1124/jpet.112.202622. Epub 2013 Jun 7. J Pharmacol Exp Ther. 2013. PMID: 23750021 Free PMC article.
-
Comparison of biological properties of 99mTc-labeled cyclic RGD Peptide trimer and dimer useful as SPECT radiotracers for tumor imaging.Nucl Med Biol. 2016 Nov;43(11):661-669. doi: 10.1016/j.nucmedbio.2016.02.006. Epub 2016 Feb 27. Nucl Med Biol. 2016. PMID: 27556955 Free PMC article.
References
-
- Weber WA, Haubner R, Vabuliene E. et al. Tumor angiogenesis targeting using imaging agents. Q J Nucl Med. 2001;45:179–82. - PubMed
-
- Liu S, Edwards DS. Fundamentals of receptor-based diagnostic metalloradiopharmaceuticals. Top Curr Chem. 2002;222:259–78.
-
- Van de Wiele C, Oltenfreiter R, De Winter O. et al. Tumor angiogenesis pathways: related clinical issues and implications for nuclear medicine imaging. Eur J Nucl Med. 2002;29:699–709. - PubMed
-
- Liu S, Robinson SP, Edwards DS. Integrin αvβ3 directed radiopharmaceuticals for tumor imaging. Drugs Fut. 2003;28:551–64.
-
- Haubner R, Wester HJ. Radiolabeled tracers for imaging of tumor angiogenesis and evaluation of anti-angiogenic therapies. Curr Pharm Des. 2004;10:1439–55. - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

