The broadly conserved regulator PhoP links pathogen virulence and membrane potential in Escherichia coli
- PMID: 21854465
- PMCID: PMC3188958
- DOI: 10.1111/j.1365-2958.2011.07804.x
The broadly conserved regulator PhoP links pathogen virulence and membrane potential in Escherichia coli
Abstract
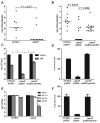
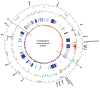
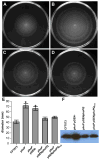
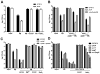
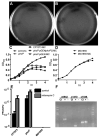
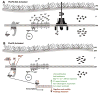
PhoP is considered a virulence regulator despite being conserved in both pathogenic and non-pathogenic Enterobacteriaceae. While Escherichia coli strains represent non-pathogenic commensal isolates and numerous virulent pathotypes, the PhoP virulence regulator has only been studied in commensal E. coli. To better understand how conserved transcription factors contribute to virulence, we characterized PhoP in pathogenic E. coli. Deletion of phoP significantly attenuated E. coli during extraintestinal infection. This was not surprising since we demonstrated that PhoP differentially regulated the transcription of > 600 genes. In addition to survival at acidic pH and resistance to polymyxin, PhoP was required for repression of motility and oxygen-independent changes in the expression of primary dehydrogenase and terminal reductase respiratory chain components. All phenotypes have in common a reliance on an energized membrane. Thus, we hypothesized that PhoP mediates these effects by regulating genes encoding proteins that generate proton motive force. Indeed, bacteria lacking PhoP exhibited a hyperpolarized membrane and dissipation of the transmembrane electrochemical gradient increased susceptibility of the phoP mutant to acidic pH, while inhibiting respiratory generation of the proton gradient restored resistance to antimicrobial peptides independent of lipopolysaccharide modification. These findings demonstrate a connection between PhoP, virulence and the energized state of the membrane.
© 2011 Blackwell Publishing Ltd.
Figures
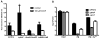
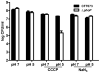
References
-
- Babior BM. Oxygen-dependent microbial killing by phagocytes (first of two parts) N Engl J Med. 1978;298:659–668. - PubMed
-
- Bader MW, Navarre WW, Shiau W, Nikaido H, Frye JG, McClelland M, Fang FC, Miller SI. Regulation of Salmonella typhimurium virulence gene expression by cationic antimicrobial peptides. Mol Microbiol. 2003;50:219–230. - PubMed
-
- Bader MW, Sanowar S, Daley ME, Schneider AR, Cho U, Xu W, Klevit RE, Le Moual H, Miller SI. Recognition of antimicrobial peptides by a bacterial sensor kinase. Cell. 2005;122:461–472. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases

