Secretory glands in cercaria of the neuropathogenic schistosome Trichobilharzia regenti - ultrastructural characterization, 3-D modelling, volume and pH estimations
- PMID: 21854564
- PMCID: PMC3171358
- DOI: 10.1186/1756-3305-4-162
Secretory glands in cercaria of the neuropathogenic schistosome Trichobilharzia regenti - ultrastructural characterization, 3-D modelling, volume and pH estimations
Abstract
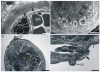
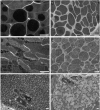
Background: Cercariae of schistosomes employ bioactive molecules for penetration into their hosts. These are released from specialized unicellular glands upon stimuli from host skin. The glands were previously well-described in the human pathogen Schistosoma mansoni. As bird schistosomes can also penetrate human skin and cause cercarial dermatitis, our aim was to characterize the architecture and ultrastructure of glands in the neurotropic bird schistosome Trichobilharzia regenti and compare it with S. mansoni. In the context of different histolytic enzymes used by these two species, we focused also on the estimations of gland volumes and pH in T. regenti.
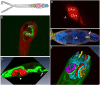
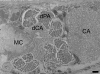
Results: The architecture and 3-D models of two types of acetabular penetration glands, their ducts and of the head gland are shown here. We characterized secretory vesicles in all three gland types by means of TEM and confirmed accuracy of the models obtained by confocal microscopy. The results of two independent approaches showed that the glands occupy ca. one third of cercarial body volume (postacetabular glands ca. 15%, circumacetabular 12% and head gland 6%). The inner environment within the two types of acetabular glands differed significantly as evidenced by dissimilar ability to bind fluorescent markers and by pH value which was higher in circumacetabular (7.44) than in postacetabular (7.08) glands.
Conclusions: As far as we know, this is the first presentation of a 3-D model of cercarial glands and the first exact estimation of the volumes of the three gland types in schistosomes. Our comparisons between T. regenti and S. mansoni implied that the architecture and ultrastructure of the glands is most likely conserved within the family. Only minor variations were found between the two species. It seems that the differences in molecular composition have no effect on general appearance of the secretory cells in TEM. Fluorescent markers employed in this study, distinguishing between secretory vesicles and gland types, can be useful in further studies of mechanisms used by cercariae for host invasion. Results of the first attempts to estimate pH within schistosome glands may help further understanding of regulation of enzymatic activities present within the glands.
Figures
References
-
- He Y, Yu Q, Yu P, Mao C, Hu Y. Penetration of Schistosoma japonicum cercaria into host skin. Chin Med J. 1990;103:34–44. - PubMed
-
- Neuhaus W. Biologie und Entwicklung von Trichobilharzia szidati n. sp. (Trematoda, Schistosomatidae), einem Erreger von Dermatitis bei Menschen. [Biology and development of Trichobilharzia szidati n. sp. (Trematoda, Schistosmatidae), a parasite causing dermatitis in man] Z Parasitenkd. 1952;15:203–266. - PubMed
-
- Müller V, Kimmig P. Trichobilharzia franki n. sp. - die Ursache fur Badedermatitiden in südwestdeutschen Baggerseen. [Trichobilharzia franki n. sp. - the cause of swimmer's dermatitis in southwest German dredged lakes] Appl Parasitol. 1994;35:12–31. - PubMed
-
- Horak P, Kolarova L, Dvorak J. Trichobilharzia regenti n. sp. (Schistosomatidae, Bilharziellinae), a new nasal schistosome from Europe. Parasite. 1998;5:349–357. - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Research Materials

