Electrophysiological correlates of sleep homeostasis in freely behaving rats
- PMID: 21854953
- PMCID: PMC3160719
- DOI: 10.1016/B978-0-444-53839-0.00002-8
Electrophysiological correlates of sleep homeostasis in freely behaving rats
Abstract
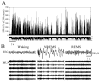
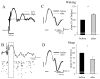
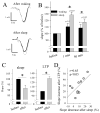
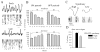
The electrical activity of the brain does not only reflect the current level of arousal, ongoing behavior, or involvement in a specific task but is also influenced by what kind of activity, and how much sleep and waking occurred before. The best marker of sleep-wake history is the electroencephalogram (EEG) spectral power in slow frequencies (slow-wave activity, 0.5-4 Hz, SWA) during sleep, which is high after extended wakefulness and low after consolidated sleep. While sleep homeostasis has been well characterized in various species and experimental paradigms, the specific mechanisms underlying homeostatic changes in brain activity or their functional significance remain poorly understood. However, several recent studies in humans, rats, and computer simulations shed light on the cortical mechanisms underlying sleep regulation. First, it was found that the homeostatic changes in SWA can be fully accounted for by the variations in amplitude and slope of EEG slow waves, which are in turn determined by the efficacy of corticocortical connectivity. Specifically, the slopes of sleep slow waves were steeper in early sleep compared to late sleep. Second, the slope of cortical evoked potentials, which is an established marker of synaptic strength, was steeper after waking, and decreased after sleep. Further, cortical long-term potentiation (LTP) was partially occluded if it was induced after a period of waking, but it could again be fully expressed after sleep. Finally, multiunit activity recordings during sleep revealed that cortical neurons fired more synchronously after waking, and less so after a period of consolidated sleep. The decline of all these electrophysiological measures-the slopes of slow waves and evoked potentials and neuronal synchrony-during sleep correlated with the decline of the traditional marker of sleep homeostasis, EEG SWA. Taken together, these data suggest that homeostatic changes in sleep EEG are the result of altered neuronal firing and synchrony, which in turn arise from changes in functional neuronal connectivity.
Copyright © 2011 Elsevier B.V. All rights reserved.
Figures
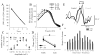

References
-
- Achermann P, Borbely AA. Low-frequency (< 1 Hz) oscillations in the human sleep electroencephalogram. Neuroscience. 1997;81:213–222. - PubMed
-
- Achermann P, Borbely AA. Temporal evolution of coherence and power in the human sleep electroencephalogram. J Sleep Res. 1998;7(Suppl 1):36–41. - PubMed
-
- Aizenman CD, Kirkwood A, Bear MF. A current source density analysis of evoked responses in slices of adult rat visual cortex: implications for the regulation of long-term potentiation. Cereb Cortex. 1996;6:751–758. - PubMed
-
- Alger BE, Teyler TJ. Long-term and short-term plasticity in the CA1, CA3, and dentate regions of the rat hippocampal slice. Brain Res. 1976;110:463–480. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources

