Onset of atrial arrhythmias elicited by autonomic modulation of rabbit sinoatrial node activity: a modeling study
- PMID: 21856904
- PMCID: PMC3774554
- DOI: 10.1152/ajpheart.00059.2011
Onset of atrial arrhythmias elicited by autonomic modulation of rabbit sinoatrial node activity: a modeling study
Abstract
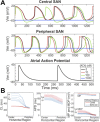
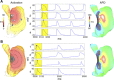
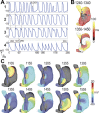
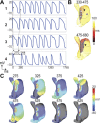
Neuronal modulation of the sinoatrial node (SAN) plays a crucial role in the initiation and maintenance of atrial arrhythmias (AF), although the exact mechanisms remain unclear. We used a computer model of a rabbit right atrium (RA) with a heterogeneous SAN and detailed ionic current descriptions for atrial and SAN myocytes to explore reentry initiation associated with autonomic activity. Heterogeneous acetylcholine (ACh)-dependent ionic responses along with L-type Ca current (I(Ca,L)) upregulation were incorporated in the SAN only. During control, activation was typical with the leading pacemaker site located close to the superior vena cava or the intercaval region. With cholinergic stimulation, activation patterns frequently included caudal shifts of the leading pacemaker site and occasional double breakouts. The model became increasingly arrhythmogenic for the ACh concentration >20 nM and for large I(Ca,L) conductance. Reentries obtained included counterclockwise rotors in the free wall, clockwise reentry circulating between the SAN and free wall, and typical flutter. The SAN was the cause of reentry with a common leading sequence of events: a bradycardic beat with shifting in the caudal direction, followed by a premature beat or unidirectional block within the SAN. Electrotonic loading, and not just overdrive pacing, squelches competing pacemaker sites in the SAN. Cholinergic stimulation concomitant with I(Ca,L) upregulation shifts leading pacemaker site and can lead to reentry. A heterogeneous response to autonomic innervation, a large myocardial load, and an extensive SAN in the intercaval region are required for neurally induced SAN-triggered reentry.
Figures



Similar articles
-
Complex interactions between the sinoatrial node and atrium during reentrant arrhythmias in the canine heart.Circulation. 2010 Aug 24;122(8):782-9. doi: 10.1161/CIRCULATIONAHA.109.935288. Epub 2010 Aug 9. Circulation. 2010. PMID: 20697021 Free PMC article.
-
Sinoatrial node reentry in a canine chronic left ventricular infarct model: role of intranodal fibrosis and heterogeneity of refractoriness.Circ Arrhythm Electrophysiol. 2013 Oct;6(5):984-94. doi: 10.1161/CIRCEP.113.000404. Epub 2013 Aug 19. Circ Arrhythm Electrophysiol. 2013. PMID: 23960214 Free PMC article.
-
Calsequestrin 2 deletion causes sinoatrial node dysfunction and atrial arrhythmias associated with altered sarcoplasmic reticulum calcium cycling and degenerative fibrosis within the mouse atrial pacemaker complex1.Eur Heart J. 2015 Mar 14;36(11):686-97. doi: 10.1093/eurheartj/eht452. Epub 2013 Nov 11. Eur Heart J. 2015. PMID: 24216388 Free PMC article.
-
The role of the calcium and the voltage clocks in sinoatrial node dysfunction.Yonsei Med J. 2011 Mar;52(2):211-9. doi: 10.3349/ymj.2011.52.2.211. Yonsei Med J. 2011. PMID: 21319337 Free PMC article. Review.
-
Three-dimensional functional anatomy of the human sinoatrial node for epicardial and endocardial mapping and ablation.Heart Rhythm. 2023 Jan;20(1):122-133. doi: 10.1016/j.hrthm.2022.08.039. Epub 2022 Sep 14. Heart Rhythm. 2023. PMID: 36113768 Free PMC article. Review.
Cited by
-
Computational assessment of the functional role of sinoatrial node exit pathways in the human heart.PLoS One. 2017 Sep 5;12(9):e0183727. doi: 10.1371/journal.pone.0183727. eCollection 2017. PLoS One. 2017. PMID: 28873427 Free PMC article.
-
Left atrial conduction times and regional velocities in persistent atrial fibrillation patients with and without fibrotic atrial cardiomyopathy.Heart Vessels. 2023 Oct;38(10):1277-1287. doi: 10.1007/s00380-023-02282-5. Epub 2023 Jul 7. Heart Vessels. 2023. PMID: 37418015 Free PMC article.
-
Mechanistic Insights Into the Reduced Pacemaking Rate of the Rabbit Sinoatrial Node During Postnatal Development: A Simulation Study.Front Physiol. 2020 Nov 20;11:547577. doi: 10.3389/fphys.2020.547577. eCollection 2020. Front Physiol. 2020. PMID: 33329016 Free PMC article.
-
Neuromodulation therapy for atrial fibrillation.Heart Rhythm. 2023 Jan;20(1):100-111. doi: 10.1016/j.hrthm.2022.08.011. Epub 2022 Aug 18. Heart Rhythm. 2023. PMID: 35988908 Free PMC article. Review.
-
Mechanistic insight into the functional role of human sinoatrial node conduction pathways and pacemaker compartments heterogeneity: A computer model analysis.PLoS Comput Biol. 2023 Dec 18;19(12):e1011708. doi: 10.1371/journal.pcbi.1011708. eCollection 2023 Dec. PLoS Comput Biol. 2023. PMID: 38109436 Free PMC article.
References
-
- Abramochkin D, Kuzmin VS, Sukhova G, Rosenshtraukh L. Modulation of rabbit sinoatrial node activation sequence by acetylcholine and isoproterenol investigated with optical mapping technique. Acta Physiol 196: 385–394, 2009. - PubMed
-
- Armour JA, Richer LP, Pagé P, Vinet A, Kus T, Vermeulen M, Nadeau R, Cardinal R. Origin and pharmacological response of atrial tachyarrhythmias induced by activation of mediastinal nerves in canines. Auton Neurosci 118: 68–78, 2005. - PubMed
-
- Beau SL, Hand DE, Schuessler RB, Bromberg BI, Kwon B, Boineau JP, Saffitz JE. Relative densities of muscarinic cholinergic and beta-adrenergic receptors in the canine sinoatrial node and their relation to sites of pacemaker activity. Circ Res 77: 957–963, 1995. - PubMed
-
- Bleeker WK, Mackaay AJ, Masson-Pévet M, Bouman LN, Becker AE. Functional and morphological organization of the rabbit sinus node. Circ Res 46: 11–22, 1980. - PubMed
-
- Boineau JP, Canavan TE, Schuessler RB, Cain ME, Corr PB, Cox JL. Demonstration of a widely distributed atrial pacemaker complex in the human heart. Circulation 77: 1221–1237, 1988. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

