Polymeric vectors for ocular gene delivery
- PMID: 21858246
- PMCID: PMC3156448
- DOI: 10.4155/tde.11.20
Polymeric vectors for ocular gene delivery
Abstract
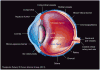
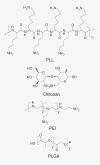
Gene therapy holds promise for the treatment of many inherited and acquired diseases of the eye. Successful ocular gene therapy interventions depend on efficient gene transfer to targeted cells with minimal toxicity. A major challenge is to overcome both intracellular and extracellular barriers associated with ocular gene delivery. Numerous viral and nonviral vectors were explored to improve transfection efficiency. Among nonviral delivery systems, polymeric vectors have gained significant attention in recent years owing to their nontoxic and non-immunogenic nature. Polyplexes or nanoparticles can be prepared by interaction of cationic polymers with DNA, which facilitate cellular uptake, endolysosomal escape and nuclear entry through active mechanisms. Chemical modification of these polymers allows for the generation of flexible delivery vectors with desirable properties. In this article several synthetic and natural polymeric systems utilized for ocular gene delivery are discussed.
Figures
Similar articles
-
Advances in polymeric and inorganic vectors for nonviral nucleic acid delivery.Ther Deliv. 2011 Apr;2(4):493-521. doi: 10.4155/tde.11.14. Ther Deliv. 2011. PMID: 22826857 Free PMC article. Review.
-
Polymer-coated viral vectors: hybrid nanosystems for gene therapy.J Gene Med. 2018 Apr;20(4):e3011. doi: 10.1002/jgm.3011. Epub 2018 Mar 25. J Gene Med. 2018. PMID: 29423922 Review.
-
[Advances in cationic polymers used as nonviral vectors for gene delivery].Sheng Wu Gong Cheng Xue Bao. 2013 May;29(5):568-77. Sheng Wu Gong Cheng Xue Bao. 2013. PMID: 24010355 Review. Chinese.
-
Hurdles to healing: Overcoming cellular barriers for viral and nonviral gene therapy.Int J Pharm. 2025 Apr 15;674:125470. doi: 10.1016/j.ijpharm.2025.125470. Epub 2025 Mar 18. Int J Pharm. 2025. PMID: 40112901 Review.
-
Recent advances in nonviral vectors for gene delivery.Acc Chem Res. 2012 Jul 17;45(7):971-9. doi: 10.1021/ar200151m. Epub 2011 Aug 26. Acc Chem Res. 2012. PMID: 21870813 Free PMC article.
Cited by
-
Therapeutic advances of miRNAs: A preclinical and clinical update.J Adv Res. 2020 Aug 29;28:127-138. doi: 10.1016/j.jare.2020.08.012. eCollection 2021 Feb. J Adv Res. 2020. PMID: 33364050 Free PMC article. Review.
-
Cell Penetrating Peptide Conjugated Chitosan for Enhanced Delivery of Nucleic Acid.Int J Mol Sci. 2015 Dec 4;16(12):28912-30. doi: 10.3390/ijms161226142. Int J Mol Sci. 2015. PMID: 26690119 Free PMC article. Review.
-
Correction of Monogenic and Common Retinal Disorders with Gene Therapy.Genes (Basel). 2017 Jan 27;8(2):53. doi: 10.3390/genes8020053. Genes (Basel). 2017. PMID: 28134823 Free PMC article. Review.
-
Topical Application of RNAi Therapy Using Surface-Modified Liposomes for Treating Retinal-Vein Occlusion.Molecules. 2025 Jun 17;30(12):2622. doi: 10.3390/molecules30122622. Molecules. 2025. PMID: 40572585 Free PMC article.
-
Immune Responses to Gene Editing by Viral and Non-Viral Delivery Vectors Used in Retinal Gene Therapy.Pharmaceutics. 2022 Sep 19;14(9):1973. doi: 10.3390/pharmaceutics14091973. Pharmaceutics. 2022. PMID: 36145721 Free PMC article. Review.
References
-
- Wu TL, Ertl HC. Immune barriers to successful gene therapy. Trends Mol Med. 2009;15:32–39. - PubMed
-
- Chaum E, Hatton MP. Gene therapy for genetic and acquired retinal diseases. Surv Ophthalmol. 2002;47:449–469. - PubMed
-
- Mohan RR, Sharma A, Netto MV, Sinha S, Wilson SE. Gene therapy in the cornea. Prog Retin Eye Res. 2005;24:537–559. - PubMed
-
- Acland GM, Aguirre GD, Ray J, et al. Gene therapy restores vision in a canine model of childhood blindness. Nat Genet. 2001;28:92–95. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical