Type I interferon and systemic lupus erythematosus
- PMID: 21859344
- PMCID: PMC3216059
- DOI: 10.1089/jir.2011.0045
Type I interferon and systemic lupus erythematosus
Abstract
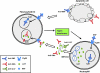
Systemic lupus erythematosus (SLE) is a complex systemic autoimmune disease associated with multiple immunologic abnormalities. Prominent among these is upregulation of type I interferon (IFN)?a powerful immune adjuvant. IFN is, in part, produced in SLE in response to autoantigens in the form of self-nucleic acids and their associated nuclear proteins. Sources of these autoantigens include apoptotic and necrotic cells as well as neutrophils undergoing a specific form of cell death called NETosis. Although plasmacytoid dendritic cells are the main producers of IFN-a, other cells are important regulators of this process. Both genetic and environmental risk factors play a role in the development and pathogenesis of SLE. Further highlighting the importance of IFN, candidate gene and genome-wide association studies have identified a number of genes involved in type I IFN pathways associated with SLE. In this review, 3 monogenic deficiencies that result in lupus-like phenotypes and several polygenic variants that have been consistently associated with SLE are highlighted, and the relationship of these genes to IFN-a production is discussed. Clinical associations of the type I IFN pathway and the use of IFN-blocking agents as therapeutic agents in SLE are also reviewed.
Figures


References
-
- Andersson G. Ek-Rylander B. Hollberg K. Ljusberg-Sjolander J. Lang P. Norgard M. Wang Y. Zhang SJ. TRACP as an osteopontin phosphatase. J Bone Miner Res. 2003;18(10):1912–1915. - PubMed
-
- Baccala R. Hoebe K. Kono DH. Beutler B. Theofilopoulos AN. TLR-dependent and TLR-independent pathways of type I interferon induction in systemic autoimmunity. Nat Med. 2007;13(5):543–551. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

