Highly practical copper(I)/TEMPO catalyst system for chemoselective aerobic oxidation of primary alcohols
- PMID: 21861488
- PMCID: PMC3197761
- DOI: 10.1021/ja206230h
Highly practical copper(I)/TEMPO catalyst system for chemoselective aerobic oxidation of primary alcohols
Abstract
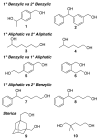
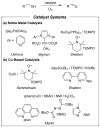
Aerobic oxidation reactions have been the focus of considerable attention, but their use in mainstream organic chemistry has been constrained by limitations in their synthetic scope and by practical factors, such as the use of pure O(2) as the oxidant or complex catalyst synthesis. Here, we report a new (bpy)Cu(I)/TEMPO catalyst system that enables efficient and selective aerobic oxidation of a broad range of primary alcohols, including allylic, benzylic, and aliphatic derivatives, to the corresponding aldehydes using readily available reagents, at room temperature with ambient air as the oxidant. The catalyst system is compatible with a wide range of functional groups and the high selectivity for 1° alcohols enables selective oxidation of diols that lack protecting groups.
Figures
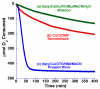
 , green); (b) “Semmelhack conditions” (similar to Table 1, entry 1): CuCl (10 mol %) and TEMPO (10 mol %) in DMF (0.4 M) (
, green); (b) “Semmelhack conditions” (similar to Table 1, entry 1): CuCl (10 mol %) and TEMPO (10 mol %) in DMF (0.4 M) ( , red); and (c) Cu(OTf) (5 mol %), bpy (5 mol %), TEMPO (5 mol %), and NMI (10 mol %) in MeCN (0.2 M) (
, red); and (c) Cu(OTf) (5 mol %), bpy (5 mol %), TEMPO (5 mol %), and NMI (10 mol %) in MeCN (0.2 M) ( , blue). See the Supporting Information for additional details.
, blue). See the Supporting Information for additional details.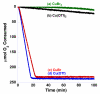
 , green), (b) Cu(OTf)2 (
, green), (b) Cu(OTf)2 ( , black), and (c) CuBr (
, black), and (c) CuBr ( , red), and (d) Cu(OTf) (
, red), and (d) Cu(OTf) ( , blue). Reactions employing CuBr2 consistently exhibit a long induction period. See the Supporting Information for additional details.
, blue). Reactions employing CuBr2 consistently exhibit a long induction period. See the Supporting Information for additional details.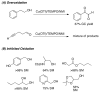
Similar articles
-
Copper(I)/ABNO-catalyzed aerobic alcohol oxidation: alleviating steric and electronic constraints of Cu/TEMPO catalyst systems.J Am Chem Soc. 2013 Oct 23;135(42):15742-5. doi: 10.1021/ja409241h. Epub 2013 Oct 15. J Am Chem Soc. 2013. PMID: 24128057 Free PMC article.
-
Mechanism of copper(I)/TEMPO-catalyzed aerobic alcohol oxidation.J Am Chem Soc. 2013 Feb 13;135(6):2357-67. doi: 10.1021/ja3117203. Epub 2013 Jan 31. J Am Chem Soc. 2013. PMID: 23317450 Free PMC article.
-
Copper(I)/TEMPO-catalyzed aerobic oxidation of primary alcohols to aldehydes with ambient air.Nat Protoc. 2012 May 24;7(6):1161-6. doi: 10.1038/nprot.2012.057. Nat Protoc. 2012. PMID: 22635108 Free PMC article.
-
Practical aerobic oxidations of alcohols and amines with homogeneous copper/TEMPO and related catalyst systems.Angew Chem Int Ed Engl. 2014 Aug 18;53(34):8824-38. doi: 10.1002/anie.201403110. Epub 2014 Jul 7. Angew Chem Int Ed Engl. 2014. PMID: 25044821 Free PMC article. Review.
-
Green, catalytic oxidations of alcohols.Acc Chem Res. 2002 Sep;35(9):774-81. doi: 10.1021/ar010075n. Acc Chem Res. 2002. PMID: 12234207 Review.
Cited by
-
Redox-Neutral TEMPO Catalysis: Direct Radical (Hetero)Aryl C-H Di- and Trifluoromethoxylation.Angew Chem Int Ed Engl. 2020 Nov 23;59(48):21475-21480. doi: 10.1002/anie.202009490. Epub 2020 Sep 24. Angew Chem Int Ed Engl. 2020. PMID: 32830430 Free PMC article.
-
Probing Cu(I) in homogeneous catalysis using high-energy-resolution fluorescence-detected X-ray absorption spectroscopy.Chem Commun (Camb). 2015 Jun 18;51(48):9864-7. doi: 10.1039/c5cc01827g. Chem Commun (Camb). 2015. PMID: 25994112 Free PMC article.
-
Dynamic self-assembly of supramolecular catalysts from precision macromolecules.Chem Sci. 2023 Aug 16;14(35):9283-9292. doi: 10.1039/d3sc03133k. eCollection 2023 Sep 13. Chem Sci. 2023. PMID: 37712032 Free PMC article.
-
Copper/TEMPO Redox Redux: Analysis of PCET Oxidation of TEMPOH by Copper(II) and the Reaction of TEMPO with Copper(I).Inorg Chem. 2019 Aug 5;58(15):10194-10200. doi: 10.1021/acs.inorgchem.9b01326. Epub 2019 Jul 8. Inorg Chem. 2019. PMID: 31283193 Free PMC article.
-
Catalytic Oxidations in a Bio-Based Economy.Front Chem. 2020 Feb 28;8:132. doi: 10.3389/fchem.2020.00132. eCollection 2020. Front Chem. 2020. PMID: 32181241 Free PMC article. Review.
References
-
- Tojo G, Fernández M. Basic Reactions in Organic Synthesis. Springer; New York: 2010. Oxidation of Alcohols to Aldehydes and Ketones.
- Tojo G, Fernández M. Basic Reactions in Organic Synthesis. Spriner; New York: 2010. Oxidation of Primary Alcohols to Carboxylic Acids.
-
- Bowden K, Heilbron IM, Jones ERH, Weedon BC. J. Chem. Soc. 1946:39–45.
- Corey EJ, Suggs JW. Tetrahedron Lett. 1975;16:2647–2650.
- Piancatelli G, Scettri A, D’Auria M. Synthesis. 1982:245–258.
- De A. J. Sci. Ind. Res. 1982;41:484–494.
- Luzzio FA, Guziec FS. Org. Prep. Proced. Int. 1988;20:533–584.
-
- Ladbury JW, Cullis CF. Chem Rev. 1958;58:403–438.
- Fatiadi AJ. Synthesis. 1976:65–104.
- Taylor RJK, Reid M, Foot J, Raw SA. Acc. Chem. Res. 2005;38:851–869. - PubMed
-
- Pfitzner KE, Moffatt JG. J. Am. Chem. Soc. 1963;85:3027–3028.
- Mancuso AJ, Huang S-L, Swern D. J. Org. Chem. 1978;43:2480–2482.
- Mancuso AJ, Brownfain DS, Swern D. J. Org. Chem. 1979;44:4148–4150.
- Tidwell TT. Synthesis. 1990:857–870.
-
- Dess DB, Martin JC. J. Org. Chem. 1983;48:4155–4156.
- Frigerio M, Santagostino M. Tetrahedron Lett. 1994;35:8019–8022.
- Uyanik M, Ishihara K. Chem. Commun. 2009:2086–2099. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

