Lef1 is required for progenitor cell identity in the zebrafish lateral line primordium
- PMID: 21862556
- PMCID: PMC3160089
- DOI: 10.1242/dev.062554
Lef1 is required for progenitor cell identity in the zebrafish lateral line primordium
Abstract
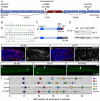
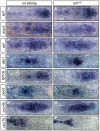
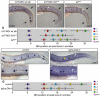
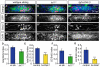
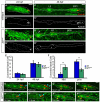
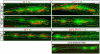
The zebrafish posterior lateral line (pLL) is a sensory system that comprises clusters of mechanosensory organs called neuromasts (NMs) that are stereotypically positioned along the surface of the trunk. The NMs are deposited by a migrating pLL primordium, which is organized into polarized rosettes (proto-NMs). During migration, mature proto-NMs are deposited from the trailing part of the primordium, while progenitor cells in the leading part give rise to new proto-NMs. Wnt signaling is active in the leading zone of the primordium and global Wnt inactivation leads to dramatic disorganization of the primordium and a loss of proto-NM formation. However, the exact cellular events that are regulated by the Wnt pathway are not known. We identified a mutant strain, lef1(nl2), that contains a lesion in the Wnt effector gene lef1. lef1(nl2) mutants lack posterior NMs and live imaging reveals that rosette renewal fails during later stages of migration. Surprisingly, the overall primordium patterning, as assayed by the expression of various markers, appears unaltered in lef1(nl2) mutants. Lineage tracing and mosaic analyses revealed that the leading cells (presumptive progenitors) move out of the primordium and are incorporated into NMs; this results in a decrease in the number of proliferating progenitor cells and eventual primordium disorganization. We concluded that Lef1 function is not required for initial primordium organization or migration, but is necessary for proto-NM renewal during later stages of pLL formation. These findings revealed a novel role for the Wnt signaling pathway during mechanosensory organ formation in zebrafish.
Figures


Similar articles
-
Lef1-dependent Wnt/β-catenin signalling drives the proliferative engine that maintains tissue homeostasis during lateral line development.Development. 2011 Sep;138(18):3931-41. doi: 10.1242/dev.062695. Development. 2011. PMID: 21862557 Free PMC article.
-
FGF-dependent mechanosensory organ patterning in zebrafish.Science. 2008 Jun 27;320(5884):1774-7. doi: 10.1126/science.1156547. Science. 2008. PMID: 18583612
-
Lef1 regulates Dusp6 to influence neuromast formation and spacing in the zebrafish posterior lateral line primordium.Development. 2013 Jun;140(11):2387-97. doi: 10.1242/dev.091348. Epub 2013 May 1. Development. 2013. PMID: 23637337 Free PMC article.
-
A framework for understanding morphogenesis and migration of the zebrafish posterior Lateral Line primordium.Mech Dev. 2017 Dec;148:69-78. doi: 10.1016/j.mod.2017.04.005. Epub 2017 Apr 28. Mech Dev. 2017. PMID: 28460893 Free PMC article. Review.
-
Signaling pathways regulating zebrafish lateral line development.Curr Biol. 2009 May 12;19(9):R381-6. doi: 10.1016/j.cub.2009.03.057. Curr Biol. 2009. PMID: 19439264 Review.
Cited by
-
Lef1-dependent hypothalamic neurogenesis inhibits anxiety.PLoS Biol. 2017 Aug 24;15(8):e2002257. doi: 10.1371/journal.pbio.2002257. eCollection 2017 Aug. PLoS Biol. 2017. PMID: 28837622 Free PMC article.
-
Guided genetic screen to identify genes essential in the regeneration of hair cells and other tissues.NPJ Regen Med. 2018 Jun 4;3:11. doi: 10.1038/s41536-018-0050-7. eCollection 2018. NPJ Regen Med. 2018. PMID: 29872546 Free PMC article.
-
Developmental disorders of the dentition: an update.Am J Med Genet C Semin Med Genet. 2013 Nov;163C(4):318-32. doi: 10.1002/ajmg.c.31382. Epub 2013 Oct 4. Am J Med Genet C Semin Med Genet. 2013. PMID: 24124058 Free PMC article. Review.
-
Disappearing scales in carps: re-visiting Kirpichnikov's model on the genetics of scale pattern formation.PLoS One. 2013 Dec 30;8(12):e83327. doi: 10.1371/journal.pone.0083327. eCollection 2013. PLoS One. 2013. PMID: 24386179 Free PMC article.
-
Innervation is required for sense organ development in the lateral line system of adult zebrafish.Proc Natl Acad Sci U S A. 2013 Apr 2;110(14):5659-64. doi: 10.1073/pnas.1214004110. Epub 2013 Mar 18. Proc Natl Acad Sci U S A. 2013. PMID: 23509277 Free PMC article.
References
-
- Abramoff M. D., Magelhaes P. J., Ram S. J. (2004). Image processing with ImageJ. Biophotonics Int. 11, 36-42
-
- Aman A., Piotrowski T. (2008). Wnt/beta-catenin and Fgf signaling control collective cell migration by restricting chemokine receptor expression. Dev. Cell 15, 749-761 - PubMed
-
- Aman A., Nguyen M., Piotrowski T. (2010). Wnt/β-catenin dependent cell proliferation underlies segmented lateral line morphogenesis. Dev. Biol. 349, 470-482 - PubMed
-
- Andermann P., Ungos J., Raible D. W. (2002). Neurogenin1 defines zebrafish cranial sensory ganglia precursors. Dev. Biol. 251, 45-58 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases

