Lef1-dependent Wnt/β-catenin signalling drives the proliferative engine that maintains tissue homeostasis during lateral line development
- PMID: 21862557
- PMCID: PMC3160090
- DOI: 10.1242/dev.062695
Lef1-dependent Wnt/β-catenin signalling drives the proliferative engine that maintains tissue homeostasis during lateral line development
Abstract
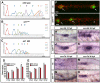
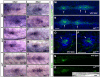
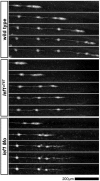
During tissue morphogenesis and differentiation, cells must self-renew while contemporaneously generating daughters that contribute to the growing tissue. How tissues achieve this precise balance between proliferation and differentiation is, in most instances, poorly understood. This is in part due to the difficulties in dissociating the mechanisms that underlie tissue patterning from those that regulate proliferation. In the migrating posterior lateral line primordium (PLLP), proliferation is predominantly localised to the leading zone. As cells emerge from this zone, they periodically organise into rosettes that subsequently dissociate from the primordium and differentiate as neuromasts. Despite this reiterative loss of cells, the primordium maintains its size through regenerative cell proliferation until it reaches the tail. In this study, we identify a null mutation in the Wnt-pathway transcription factor Lef1 and show that its activity is required to maintain proliferation in the progenitor pool of cells that sustains the PLLP as it undergoes migration, morphogenesis and differentiation. In absence of Lef1, the leading zone becomes depleted of cells during its migration leading to the collapse of the primordium into a couple of terminal neuromasts. We show that this behaviour resembles the process by which the PLLP normally ends its migration, suggesting that suppression of Wnt signalling is required for termination of neuromast production in the tail. Our data support a model in which Lef1 sustains proliferation of leading zone progenitors, maintaining the primordium size and defining neuromast deposition rate.
Figures





Similar articles
-
Lef1 is required for progenitor cell identity in the zebrafish lateral line primordium.Development. 2011 Sep;138(18):3921-30. doi: 10.1242/dev.062554. Development. 2011. PMID: 21862556 Free PMC article.
-
Lef1 regulates Dusp6 to influence neuromast formation and spacing in the zebrafish posterior lateral line primordium.Development. 2013 Jun;140(11):2387-97. doi: 10.1242/dev.091348. Epub 2013 May 1. Development. 2013. PMID: 23637337 Free PMC article.
-
Wnt/β-catenin dependent cell proliferation underlies segmented lateral line morphogenesis.Dev Biol. 2011 Jan 15;349(2):470-82. doi: 10.1016/j.ydbio.2010.10.022. Epub 2010 Oct 23. Dev Biol. 2011. PMID: 20974120
-
A framework for understanding morphogenesis and migration of the zebrafish posterior Lateral Line primordium.Mech Dev. 2017 Dec;148:69-78. doi: 10.1016/j.mod.2017.04.005. Epub 2017 Apr 28. Mech Dev. 2017. PMID: 28460893 Free PMC article. Review.
-
Signaling pathways regulating zebrafish lateral line development.Curr Biol. 2009 May 12;19(9):R381-6. doi: 10.1016/j.cub.2009.03.057. Curr Biol. 2009. PMID: 19439264 Review.
Cited by
-
Organized emergence of multiple-generations of teeth in snakes is dysregulated by activation of Wnt/beta-catenin signalling.PLoS One. 2013 Sep 3;8(9):e74484. doi: 10.1371/journal.pone.0074484. eCollection 2013. PLoS One. 2013. PMID: 24019968 Free PMC article.
-
A Hox gene controls lateral line cell migration by regulating chemokine receptor expression downstream of Wnt signaling.Proc Natl Acad Sci U S A. 2013 Oct 15;110(42):16892-7. doi: 10.1073/pnas.1306282110. Epub 2013 Sep 30. Proc Natl Acad Sci U S A. 2013. PMID: 24082091 Free PMC article.
-
Yap and Taz regulate retinal pigment epithelial cell fate.Development. 2015 Sep 1;142(17):3021-32. doi: 10.1242/dev.119008. Epub 2015 Jul 24. Development. 2015. PMID: 26209646 Free PMC article.
-
Generation and application of signaling pathway reporter lines in zebrafish.Mol Genet Genomics. 2013 Jun;288(5-6):231-42. doi: 10.1007/s00438-013-0750-z. Epub 2013 May 15. Mol Genet Genomics. 2013. PMID: 23674148 Free PMC article. Review.
-
Loss of vacuolar-type H+-ATPase induces caspase-independent necrosis-like death of hair cells in zebrafish neuromasts.Dis Model Mech. 2021 Jul 1;14(7):dmm048997. doi: 10.1242/dmm.048997. Epub 2021 Jul 23. Dis Model Mech. 2021. PMID: 34296747 Free PMC article.
References
-
- Aman A., Piotrowski T. (2008). Wnt/beta-catenin and Fgf signaling control collective cell migration by restricting chemokine receptor expression. Dev. Cell 15, 749-761 - PubMed
-
- Aman A., Nguyen M., Piotrowski T. (2011). Wnt/beta-catenin dependent cell proliferation underlies segmented lateral line morphogenesis. Dev. Biol. 349, 470-482 - PubMed
-
- Clevers H. (2006). Wnt/beta-catenin signaling in development and disease. Cell 127, 469-480 - PubMed
-
- Collazo A., Fraser S. E., Mabee P. M. (1994). A dual embryonic origin for vertebrate mechanoreceptors. Science 264, 426-430 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials

