Crosstalk between MYCN and MDM2-p53 signal pathways regulates tumor cell growth and apoptosis in neuroblastoma
- PMID: 21862876
- PMCID: PMC3218600
- DOI: 10.4161/cc.10.17.17118
Crosstalk between MYCN and MDM2-p53 signal pathways regulates tumor cell growth and apoptosis in neuroblastoma
Abstract
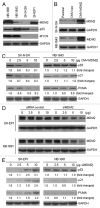
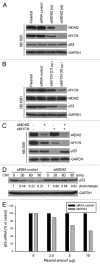
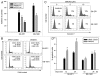
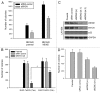
Previous studies show that the MYCN and MDM2-p53 signal pathways are mutually regulated: MYCN stimulates MDM2 and p53 transcription, whereas MDM2 stabilizes MYCN mRNA and induces its translation. Herein, we report that the interaction between MDM2 and MYCN plays a critical role in MYCN-amplified neuroblastoma tumor cell growth and survival. Distinct from the known role that MDM2 has in regulating tumor promotion in non-MYCN-amplified neuroblastoma, in which MDM2 inhibits p53, we found that MDM2 stimulated tumor growth in MYCN-amplified neuroblastoma in a p53-independent manner. In MYCN-amplified neuroblastoma cells, enforced expression of MDM2 further enhanced MYCN expression, yet no p53 inhibition was observed by MDM2 due to upregulation of MYCN that stimulated p53 transcription. Similarly, p53 expression remained unchanged in MDM2-silenced MYCN-amplified neuroblastoma cells because MDM2 inhibition resulted in a downregulation of MYCN that decreased p53 transcription, although the MDM2-mediated degradation of p53 was reduced. Also, we found that the enforced overexpression of MDM2, or conversely, the inhibition of overexpressed endogenous MDM2, led to either a remarkable increase or decrease in tumor growth, respectively, in MYCN-amplified neuroblastoma (even though no p53 function was involved). These results suggest that p53 that is reciprocally regulated by MDM2 and MYCN is dispensable for suppression of MYCN-amplified neuroblastoma, and that the direct interaction between MDM2 and MYCN may contribute significantly to MYCN-amplified neuroblastoma growth and disease progression.
© 2011 Landes Bioscience
Figures

References
-
- Maris JM, Matthay KK. Molecular biology of neuroblastoma. J Clin Oncol. 1999;17:2264–2279. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous
