Supramolecular coordination: self-assembly of finite two- and three-dimensional ensembles
- PMID: 21863792
- PMCID: PMC3212633
- DOI: 10.1021/cr200077m
Supramolecular coordination: self-assembly of finite two- and three-dimensional ensembles
Figures

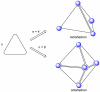
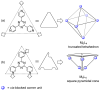
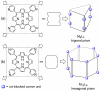

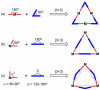

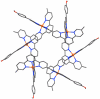
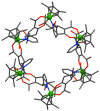
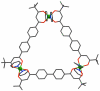
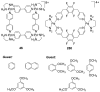
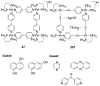
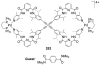
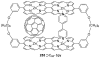

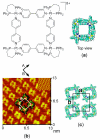


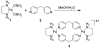
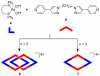
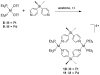
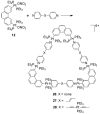
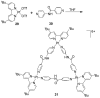
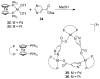
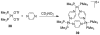

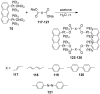
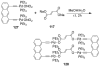
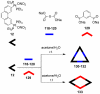
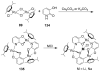
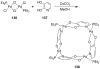
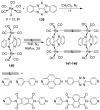
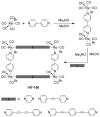
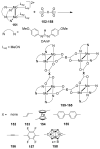

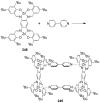

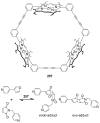
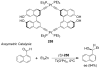
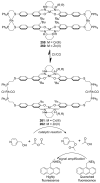

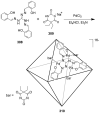
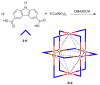
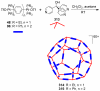
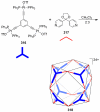
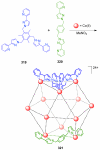
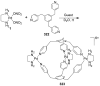
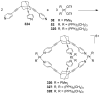
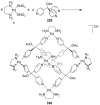
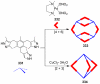
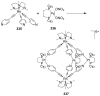
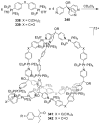
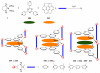
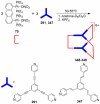
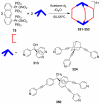
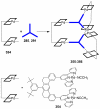
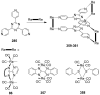
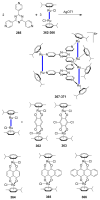
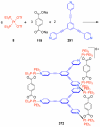
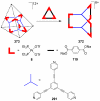
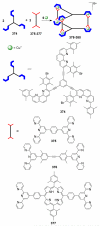
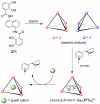
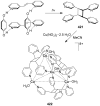


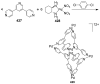
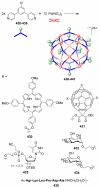
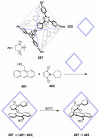
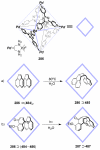
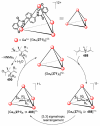
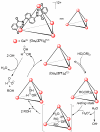
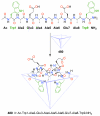
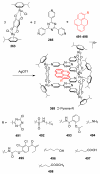
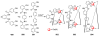
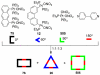
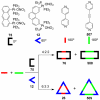
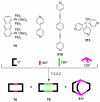
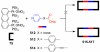
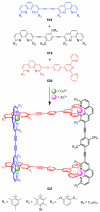
References
-
- Pedersen CJ. Angew. Chem. Int. Ed. Engl. 1988;27:1021.
-
- Lehn J-M. Angew. Chem. Int. Ed. Engl. 1988;27:89.
-
- Cram DJ. Angew. Chem. Int. Ed. Engl. 1988;27:1009.
-
- Steed JW, Turner DR, Wallace KJ. Core Concepts in Supramolecular Chemistry and Nanochemistry. John Wiley & Sons; West Sussex: 2007.
- Steed JW, Atwood JL. Supramolecular Chemistry. John Wiley & Sons; West Sussex: 2000.
-
- Lehn J-M. Supramolecular Chemistry: Concepts and Perspectives. VCH; Weinheim: 1995.
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

