Broad and potent immune responses to a low dose intradermal HIV-1 DNA boosted with HIV-1 recombinant MVA among healthy adults in Tanzania
- PMID: 21864626
- PMCID: PMC4795940
- DOI: 10.1016/j.vaccine.2011.08.001
Broad and potent immune responses to a low dose intradermal HIV-1 DNA boosted with HIV-1 recombinant MVA among healthy adults in Tanzania
Abstract
Background: We conducted a phase I/II randomized placebo-controlled trial with the aim of exploring whether priming with a low intradermal dose of a multiclade, multigene HIV-1 DNA vaccine could improve the immunogenicity of the same vaccine given intramuscularly prior to boosting with a heterologous HIV-1 MVA among healthy adults in Dar es Salaam, Tanzania.
Methods: Sixty HIV-uninfected volunteers were randomized to receive DNA plasmid vaccine 1mg intradermally (id), n=20, or 3.8mg intramuscularly (im), n=20, or placebo, n=20, using a needle-free injection device. DNA plasmids encoding HIV-1 genes gp160 subtype A, B, C; rev B; p17/p24 gag A, B and Rtmut B were given at weeks 0, 4 and 12. Recombinant MVA (10(8)pfu) expressing HIV-1 Env, Gag, Pol of CRF01_AE or placebo was administered im at month 9 and 21.
Results: The vaccines were well tolerated. Two weeks after the third HIV-DNA injection, 22/38 (58%) vaccinees had IFN-γ ELISpot responses to Gag. Two weeks after the first HIV-MVA boost all 35 (100%) vaccinees responded to Gag and 31 (89%) to Env. Two to four weeks after the second HIV-MVA boost, 28/29 (97%) vaccinees had IFN-γ ELISpot responses, 27 (93%) to Gag and 23 (79%) to Env. The id-primed recipients had significantly higher responses to Env than im recipients. Intracellular cytokine staining for Gag-specific IFN-γ/IL-2 production showed both CD8(+) and CD4(+) T cell responses. All vaccinees had HIV-specific lymphoproliferative responses. All vaccinees reacted in diagnostic HIV serological tests and 26/29 (90%) had antibodies against gp160 after the second HIV-MVA boost. Furthermore, while all of 29 vaccinee sera were negative for neutralizing antibodies against clade B, C and CRF01_AE pseudoviruses in the TZM-bl neutralization assay, in a PBMC assay, the response rate ranged from 31% to 83% positives, depending upon the clade B or CRF01_AE virus tested.
Conclusions: This vaccine approach is safe and highly immunogenic. Low dose, id HIV-DNA priming elicited higher and broader cell-mediated immune responses to Env after HIV-MVA boost compared to a higher HIV-DNA priming dose given im. Three HIV-DNA priming immunizations followed by two HIV-MVA boosts efficiently induced Env-antibody responses.
Copyright © 2011 Elsevier Ltd. All rights reserved.
Conflict of interest statement
Figures
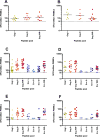



References
-
- UNAIDS. UNAIDS report on the global AIDS epidemic. 2010 Available from: www.unaids.org/globalreport/Global_report.htm.
-
- Paris RM, Kim JH, Robb ML, Michael NL. Prime-boost immunization with poxvirus or adenovirus vectors as a strategy to develop a protective vaccine for HIV-1. Expert Rev Vaccines. 2010;9(9):1055–69. - PubMed
-
- Gilbert PB, Peterson ML, Follmann D, Hudgens MG, Francis DP, Gurwith M, et al. Correlation between immunologic responses to a recombinant glycoprotein 120 vaccine and incidence of HIV-1 in a phase 3 HIV-1 preventive vaccine trial. J Infect Dis. 2005;191(5):666–77. - PubMed
-
- Robinson HL, Amara RR. T cell vaccines for microbial infections. Nat Med. 2005;11:S25–32. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

