Counting ion and water molecules in a streaming file through the open-filter structure of the K channel
- PMID: 21865461
- PMCID: PMC6623228
- DOI: 10.1523/JNEUROSCI.1377-11.2011
Counting ion and water molecules in a streaming file through the open-filter structure of the K channel
Abstract
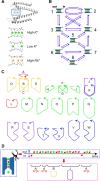
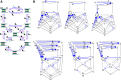
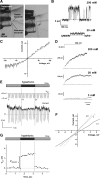
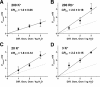
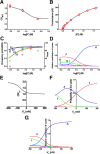
The mechanisms underlying the selective permeation of ions through channel molecules are a fundamental issue related to understanding how neurons exert their functions. The "knock-on" mechanism, in which multiple ions in the selectivity filter are hit by an incoming ion, is one of the leading concepts. This mechanism has been supported by crystallographic studies that demonstrated ion distribution in the structure of the Streptomyces lividans (KcsA) potassium channel. These still pictures under equilibrium conditions, however, do not provide a snapshot of the actual, ongoing permeation processes. To understand the dynamics of permeation, we determined the ratio of the ion and water flow [the water-ion coupling ratio (CR(w-i))] through the KcsA channel by measuring the streaming potential (V(stream)) electrophysiologically. The V(stream) value was converted to the CR(w-i) value, which reveals how individual ion and water molecules are queued in the narrow and short filter during permeation. At high K(+) concentrations, the CR(w-i) value was 1.0, indicating that turnover between the alternating ion and water arrays occurs in a single-file manner. At low K(+), the CR(w-i) value was increased to a point over 2.2, suggesting that the filter contained mostly one ion at a time. These average behaviors of permeation were kinetically analyzed for a more detailed understanding of the permeation process. Here, we envisioned the permeation as queues of ion and water molecules and sequential transitions between different patterns of arrays. Under physiological conditions, we predicted that the knock-on mechanism may not be predominant.
Figures

Similar articles
-
Filter gate closure inhibits ion but not water transport through potassium channels.Proc Natl Acad Sci U S A. 2013 Jun 25;110(26):10842-7. doi: 10.1073/pnas.1304714110. Epub 2013 Jun 10. Proc Natl Acad Sci U S A. 2013. PMID: 23754382 Free PMC article.
-
Unified modeling of conductance kinetics for low- and high-conductance potassium ion channels.Phys Rev E Stat Nonlin Soft Matter Phys. 2006 Jul;74(1 Pt 1):011902. doi: 10.1103/PhysRevE.74.011902. Epub 2006 Jul 5. Phys Rev E Stat Nonlin Soft Matter Phys. 2006. PMID: 16907122
-
Queueing arrival and release mechanism for K+ permeation through a potassium channel.J Physiol Sci. 2019 Nov;69(6):919-930. doi: 10.1007/s12576-019-00706-4. Epub 2019 Aug 27. J Physiol Sci. 2019. PMID: 31456113 Free PMC article.
-
EPR approaches to ion channel structure and function.Novartis Found Symp. 2002;245:146-58; discussion 158-64, 165-8. doi: 10.1002/0470868759.ch10. Novartis Found Symp. 2002. PMID: 12027005 Review.
-
The Persistent Question of Potassium Channel Permeation Mechanisms.J Mol Biol. 2021 Aug 20;433(17):167002. doi: 10.1016/j.jmb.2021.167002. Epub 2021 Apr 20. J Mol Biol. 2021. PMID: 33891905 Review.
Cited by
-
Channel function reconstitution and re-animation: a single-channel strategy in the postcrystal age.J Physiol. 2015 Jun 15;593(12):2553-73. doi: 10.1113/JP270025. Epub 2015 May 14. J Physiol. 2015. PMID: 25833254 Free PMC article. Review.
-
Conductance selectivity of Na+ across the K+ channel via Na+ trapped in a tortuous trajectory.Proc Natl Acad Sci U S A. 2021 Mar 23;118(12):e2017168118. doi: 10.1073/pnas.2017168118. Proc Natl Acad Sci U S A. 2021. PMID: 33741736 Free PMC article.
-
Structure, function, and ion-binding properties of a K+ channel stabilized in the 2,4-ion-bound configuration.Proc Natl Acad Sci U S A. 2019 Aug 20;116(34):16829-16834. doi: 10.1073/pnas.1901888116. Epub 2019 Aug 6. Proc Natl Acad Sci U S A. 2019. PMID: 31387976 Free PMC article.
-
The open gate structure of the membrane-embedded KcsA potassium channel viewed from the cytoplasmic side.Sci Rep. 2013;3:1063. doi: 10.1038/srep01063. Epub 2013 Jan 15. Sci Rep. 2013. PMID: 23323207 Free PMC article.
-
Aquaporin water channels in the nervous system.Nat Rev Neurosci. 2013 Apr;14(4):265-77. doi: 10.1038/nrn3468. Epub 2013 Mar 13. Nat Rev Neurosci. 2013. PMID: 23481483 Free PMC article. Review.
References
-
- Bernèche S, Roux B. Energetics of ion conduction through the K+ channel. Nature. 2001;414:73–77. - PubMed
-
- Colquhoun D, Hawkes AG. The principles of the stochastic interpretation of ion-channel mechanisms. In: Sakmann B, Neher E, editors. Single-channel recording. 2nd Ed. New York: Plenum; 1995. pp. 397–482.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
