Small molecule inhibitors reveal Niemann-Pick C1 is essential for Ebola virus infection
- PMID: 21866101
- PMCID: PMC3230319
- DOI: 10.1038/nature10380
Small molecule inhibitors reveal Niemann-Pick C1 is essential for Ebola virus infection
Abstract
Ebola virus (EboV) is a highly pathogenic enveloped virus that causes outbreaks of zoonotic infection in Africa. The clinical symptoms are manifestations of the massive production of pro-inflammatory cytokines in response to infection and in many outbreaks, mortality exceeds 75%. The unpredictable onset, ease of transmission, rapid progression of disease, high mortality and lack of effective vaccine or therapy have created a high level of public concern about EboV. Here we report the identification of a novel benzylpiperazine adamantane diamide-derived compound that inhibits EboV infection. Using mutant cell lines and informative derivatives of the lead compound, we show that the target of the inhibitor is the endosomal membrane protein Niemann-Pick C1 (NPC1). We find that NPC1 is essential for infection, that it binds to the virus glycoprotein (GP), and that antiviral compounds interfere with GP binding to NPC1. Combined with the results of previous studies of GP structure and function, our findings support a model of EboV infection in which cleavage of the GP1 subunit by endosomal cathepsin proteases removes heavily glycosylated domains to expose the amino-terminal domain, which is a ligand for NPC1 and regulates membrane fusion by the GP2 subunit. Thus, NPC1 is essential for EboV entry and a target for antiviral therapy.
Figures
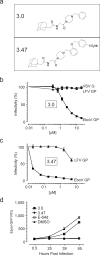
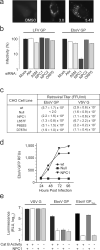

Comment in
-
Achilles heel of Ebola viral entry.Nat Rev Drug Discov. 2011 Sep 30;10(10):731. doi: 10.1038/nrd3568. Nat Rev Drug Discov. 2011. PMID: 21959282 No abstract available.
References
-
- Geisbert TW, Jahrling PB. Exotic emerging viral diseases: progress and challenges. Nat. Med. 2004;10:S110–121. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases

